Abstract
There is a need to cultivate medicinal plants to meet the growing demand. Their cultivation is hampered by extreme environmental conditions such as drought that affect plant growth and its pharmacological potential. Application of stress-tolerant endophytic species may potentially attenuate these negative impacts. This study assessed the effects of individual and co-inoculation of two native endophytic species (bacterium Paenibacillus polymyxa and fungus Fusarium oxysporum) on growth, physiological responses, metabolite accumulation and therapeutic efficacy of Endostemon obtusifolius subjected to varying watering regimes (well watered, mild and severe stress) under greenhouse conditions. Drought stress negatively affected root and shoot biomass, carotenoid content, chlorophyll fluorescence and relative water content in E. obtusifolius. Electrolyte leakage and malondialdehyde and hydrogen peroxide accumulation increased with drought stress. Individual and co-inoculation endophyte treatments significantly improved growth and stress tolerance mechanisms via increased osmolyte production (soluble sugars, proline), up-regulation of the enzymatic antioxidant system (superoxide dismutase) and increased antioxidant metabolite content (total phenolics, flavonoids). Antioxidant (DPPH, FRAP) and in vitro α-glucosidase activity of ethyl acetate leaf extracts were negatively affected by water stress but significantly improved when plantlets were subjected to endophyte inoculation. The most active extracts were from plants subjected to mild water stress with co-inoculation. Thus severe drought stress negatively affected growth and therapeutic efficacy of E. obtusifolius. Inoculation with beneficial endophytes enhanced the biochemical responses, osmoregulatory network and improved the therapeutic efficacy of E. obtusifolius.
Similar content being viewed by others
Introduction
Wild populations of medicinal herbs are under pressure due to increased harvesting for research, trade, therapeutic and industrial benefits (Seile et al. 2022). This has led to a loss of biodiversity and habitat degradation (Nwafor et al. 2021). It is thus important to develop cultivation practices to meet the escalating demand for these plants with active principles and to restore their environment. Cultivation strategies need to focus on optimum cultivation conditions, ways to enhance in-planta biosynthesis of valuable bioactive compounds and to achieve high-quality product uniformity (Alamgir 2017). Adverse climatic conditions, diseases and pests, crop failure and the high cost of cultivation are some of the global problems hindering extensive cultivation of medicinal plants (Alamgir 2017).
Drought is a major global environmental threat affecting plant growth and yield. Water deficit effects can manifest at different levels of plant organization and may impair processes such as water absorption, mineral nutrient uptake, photosynthesis, gaseous exchange, cell division and cellular respiration (Kapoor et al. 2020). This affects the health and efficiency of the plant (Caser et al. 2019). Tolerance to drought stress depends on the plant species and genotype, the timing and severity of the stress and the developmental stage of the plant (Kakar et al. 2022; Khan et al. 2018). Excessive reactive oxygen species (ROS) accumulate in plant tissues as a consequence of drought. This is lethal to cells as ROS peroxidizes membrane lipids, damages other macromolecules such as proteins and nucleic acids, inhibits enzymatic activities and ultimately leads to cell death (Khan et al. 2018). Plants activate a series of protective mechanisms in response to drought and oxidative stress. These include osmolyte (e.g. proline and soluble sugar) synthesis and accumulation, enhanced activities of antioxidant enzymes such as superoxide dismutase (SOD) and accumulation of non-enzymatic antioxidants (e.g. phenolic compounds and ascorbic acid) to scavenge toxic free radicals (Kapoor et al. 2020).
Resistance to drought and other stresses can be further enhanced by symbiont endophytes (fungi and bacteria) that colonize the plant’s tissues (Rho et al. 2018; Salvi et al. 2022). These are typically mutualistic microbes that occur inter- and intra-cellularly in plant tissue without the plant showing any infection or adverse effects (Das et al. 2023; Khare et al. 2018). Under drought conditions, endophytes, in addition to their host’s innate responses, promote growth, suppress diseases, assist in nutrient assimilation and induce drought tolerance via the synthesis of certain enzymes, exopolysaccharides, growth regulators and volatile compounds (Lu et al. 2021). They may also mediate stress-responsive genes, elicit anatomical and physiological responses and increase the in planta concentrations of antioxidants and osmolytes (Khare et al. 2018). They also promote the accumulation of bioactive compounds in medicinal plants (Yang et al. 2019).
Drought is one of the foremost environmental stress factors in arid and semi-arid regions and influences not only growth and development but also the curative properties of medicinal plants (Yue et al. 2022). Endostemon obtusifolius (E. Mey. ex Benth.) N. E. Br. of the Lamiaceae (mint) family, is a perennial shrub indigenous to southern Africa. It grows in semi-arid regions of South Africa and is thus perpetually exposed to some level of water deficit. The leaves of this aromatic plant are used traditionally as a culinary herb and by extension, as a medicinal plant (Sadashiva et al. 2014). It has neuroprotective, analgesic, anesthetic, antidiabetic and antioxidant properties (Sadashiva et al. 2014; Ogbe et al. 2023a). Plant secondary metabolites with antioxidant properties are abundantly distributed in the Mint family (Tamokou et al. 2017) and play a protective role in the pathogenesis of degenerative diseases including renal dysfunction, diabetes, obesity, high blood pressure and atherosclerosis (Ong et al. 2018).
One potential economically viable strategy to enhance the synthesis of valuable secondary metabolites is to deliberately subject cultivated medicinal plants to different abiotic stresses together with the application of exogenous substances and beneficial microorganisms (biotic elicitors) such as endophytic symbionts to improve their stress tolerance (Ouyang et al. 2023; Zhang et al. 2019) and accumulation of bioactive compounds (Yang et al. 2019). Previous studies on E. obtusifolius have mainly focused on its pharmacological value. Its morphological, biochemical and physiological responses as well as its pharmacological status under drought stress have not been studied. Previously, 26 endophytes (12 fungi and 14 bacteria) were isolated from the leaves and roots of E. obtusifolius and their growth-promoting characteristics assessed. The two most promising isolates were identified as the bacterium Paenibacillus polymyxa isolated from the roots and the fungus Fusarium oxysporum (EOLF-5) isolated from the leaves of E. obtusifolius (Ogbe et al. 2023b). The aim of the present study was to investigate the effects of individual and co-inoculations of the previously isolated P. polymyxa and F. oxysporum endophytes on the growth, drought tolerance and pharmacological activities of E. obtusifolius grown under different watering regimes.
Materials and Methods
Cultivation of Endostemon obtusifolius and Growth Conditions
Endostemon obtusifolius stem cuttings were prepared according to the procedure of Anith et al. (2018) with some modifications. Stem cuttings of approximately 10 cm long and 3 mm diameter with 3–4 nodes were harvested from young and disease-free lateral branches of a solitary mother E. obtusifolius plant growing in the University of KwaZulu-Natal Botanical Gardens, Pietermaritzburg Campus (latitude − 29.625073, longitude 30.403557). The cuttings were defoliated except for a flag leaf and placed in sterile distilled water until treated. The basal portion of the stem cuttings were dipped in a rooting hormone (Seradix B No.3, Bayer Crop Science, Germany) for 1 min and planted in disinfected trays (27 × 18 cm) containing sterile vermiculite. The trays were placed in a mist house with day and night temperatures of 28/15 ºC, 80–90% relative humidity and 10 s misting at 15 min intervals. After three weeks, the stem cuttings were transplanted into 7.5 cm diameter pots containing sterile palm peat and vermiculite (1:1) and placed in a greenhouse with 30–40% relative humidity and an average photosynthetic photon flux density of 450 µmol m−2 s−1. After three weeks in the greenhouse, well-rooted stem cuttings were transplanted into 15 cm diameter pots containing 1:3 sterile vermiculite and twice autoclaved garden soil. The physiochemical properties of the medium were 11% moisture, 5 mg kg−1 P, 1.7 mg kg−1 K, 5 mg kg−1 alkali hydrolysable N, 9 mg kg−1 Ca, 40% field capacity (FC) and pH 7.80. These were placed under the same greenhouse conditions as above and irrigated as required with half-strength Hoagland’s Solution (Hoagland and Snyder 1933) for four weeks.
Preparation and Application of Microbial Inoculants
Previously isolated and identified endophytes (Paenibacillus polymyxa and Fusarium oxysporum) from E. obtusifolius were used (Ogbe et al. 2023b). The bacterial endophyte, P. polymyxa inoculum was prepared by growing the isolate in Schott bottles containing 250 mL Mueller Hinton Broth and incubating at 28 °C on a shaking incubator at 180 rpm for two days. Subsequently, the cells in the broth were harvested by centrifugation (Avanti ® J-E Centrifuge Beckman Coulter, Ireland) at 5 000 × g for 10 min at 4 °C, washed and resuspended in sterile distilled water. The cell density was adjusted to 1 × 107 CFU mL−1 final concentration using sterile distilled water (Hussain et al. 2018). The fungal endophyte was maintained on potato dextrose agar plates for two weeks at 28 ºC in an incubator. Thereafter, the plates were flooded with 10 mL sterilized distilled water containing 0.05% (v/v) Tween-20. The fungal colonies were then gently removed from the media surface using a sterile spatula and transferred to a sterilized beaker. The conidial suspension was made by filtering the harvested mycelial mass through a double layer of sterilized cheesecloth to remove the agar and mycelia debris. The final spore number was evaluated using a haemocytometer and adjusted to 7 × 105 (Sadeghi et al. 2020).
A modified soil-drenching inoculation method of Ramakuwela et al. (2020) was employed where for single inoculation, either 20 mL bacterial suspension or 80 mL conidial suspension was poured around the root zone of the E. obtusifolius cuttings. A co-inoculation treatment was done by applying 20 mL bacterial suspension and 80 mL conidial suspension around the same region. An uninoculated control group was treated with 100 mL distilled water. To confirm the establishment of the inoculated endophytes in the stem cuttings, the roots from three pots of each sub-group were carefully harvested without disturbing the integrity of the root architecture five weeks after inoculation. The isolation procedure described in Ogbe et al. (2023b) was followed and the inoculated isolates were re-identified on plates using morphological characters.
Experimental Design and Treatments
The individual and combined effects of P. polymyxa and F. oxysporum inoculations on drought-stressed E. obtusifolius stem cuttings were investigated by setting up a factorial experiment with two combined factors: (1) inoculation with P. polymyxa, inoculation with F. oxysporum, co-inoculation with P. polymyxa and F. oxysporum and uninoculated control; (2) soil water content - well-watered with 100% FC, mild stress with 50% FC and severe stress with 25% FC. Each of the twelve treatments had ten replicates and the plants (ten-week-old stem cuttings) were arranged in a completely randomized fashion. Six weeks after inoculation (16 weeks post-stem cuttings), the plants were subjected to drought stress treatments for 8 weeks.
Determination of Dry Shoot and Root Weight
After 8 weeks drought treatment, three plants per treatment were randomly harvested, washed under running tap water and divided into root and shoot systems. The plants were oven-dried at 70 ºC for 48 h until a constant weight was reached after which the shoot and dry weights (DW) were measured.
All the following measurements were carried out on leaves harvested from three representative pots per treatment (n = 3).
Photosynthetic Pigments
Total chlorophyll (a + b) and carotenoids of fully expanded leaves of E. obtusifolius plants were determined according to the method of Lichtenthaler (1987). Leaves were weighed and homogenized in 80% acetone. The extract was then centrifuged (1,000 × g for 10 min at 0 °C; Hettich Universal, Tuttlingen, Germany) and the absorbance of the samples measured at 470, 645 and 662 nm. The chlorophyll content was calculated (mg g−1 fresh weight [FW]) using the formulae of Lichtenhaler (1987).
Photochemical Efficiency of Photosystem II (Fv/Fm)
Chlorophyll fluorescence of E. obtusifolius plants was assessed after 7 weeks of drought treatment using a portable chlorophyll fluorometer (OS-30p; Opti-Sciences, Inc., NH, USA) using leaves. Chlorophyll fluorescence measurements were carried out in the dark on the upper leaf (adaxial) surface on the youngest but mature leaf of each plant from three representative pots. After an initial 45 min dark adaptation, minimal fluorescence (F0) was recorded by applying weak modulated light (0.4 µmol m−2 s−1). Maximal fluorescence (Fm) was subsequently measured after illumination with a saturating flashlight (8000 µmol m−2 s−1) for 0.8 s. Measurements were taken in triplicate from each leaf. Variable fluorescence (Fv) was computed by an in-built programme to determine the maximal photochemical efficiency of PSII (Khan et al. 2016).
Relative Water Content
The relative water content (RWC) of the leaves was estimated as per Zhang et al. (2019) with slight modifications. Fully developed leaves were selected and their FWs immediately recorded. The leaves were then immersed in 25 mL distilled water and incubated in the dark for 24 h at 4 °C. After drying the surface water with a paper towel, the turgid weight (TW) of each leaf was recorded. The samples were then oven-dried at 70 °C for 48 h and the DW measured. The water status was evaluated by estimating RWC using the formula:
Electrolyte Leakage
Electrolyte leakage was estimated using leaf discs (0.5 cm) obtained from leaf samples to measure cell membrane stability. Briefly, leaf discs were immersed in 10 mL distilled water for 4 h at room temperature. The electrical conductivity of the medium (EC1) was recorded using an electrical conductivity meter (Laborgerate GmbH). Thereafter, the sealed samples were autoclaved at 121 °C for 15 min to disrupt the cell membrane and fully release the electrolytes. After cooling at room temperature, the final electrical conductivity (EC2) was measured. Electrolyte leakage percentage was calculated using the formula (Kumar et al. 2018):
Total Soluble Sugar Content
The total soluble sugar in E. obtusifolius leaves was estimated using the anthrone reagent method as described by Tyagi et al. (2017). Briefly, 200 mg freshly harvested leaves were hydrolyzed in 5 mL 2.5 N HCl before adding anthrone reagent. The absorbance was measured at 630 nm. The amount of total soluble sugar was estimated from a glucose concentration standard curve and expressed as µg g−1 FW.
Proline Content
The proline content in the leaves of E. obtusifolius was determined following the Bibi et al. (2019) protocol. Briefly, 100 mg fresh leaf samples were homogenized in 5 mL 3% sulfosalicylic acid. After addition of a reaction mixture (250 µL 3% sulfosalicylic acid, 500 µL glacial acetic acid and 250 µL 2.5% acidic ninhydrin), toluene was added to separate the chromophore containing organic phase. The absorbance of the organic phase was measured at 520 nm. Proline concentration was calculated from a proline standard calibration curve and expressed as mg g−1 FW.
SOD Activity
SOD activity was quantified using the nitroblue tetrazolium (NBT) method described by Ye et al. (2021). Briefly, enzyme extract was prepared by homogenizing 200 mg FW leaf samples of E. obtusifolius in 2 mL extraction solution (100 mM phosphate buffer (pH 7.8) with 0.1 mM EDTA). The prepared enzyme extract was added to a reaction mixture (130 mM methionine, 750 µM NBT, 20 µM riboflavin and 1 mM EDTANa2) and incubated under light for 30 min at room temperature. SOD activity was quantified spectrophotometrically at 560 nm. One unit of SOD activity was calculated as the amount of enzyme that suppressed the photoreduction rate of NBT by 50% compared to tubes without enzyme extracts and was expressed as SOD U g−1 FW. Protein concentration was measured using bovine serum albumin as a standard (Bradford 1976).
Malondialdehyde (MDA) Content
MDA concentration in the leaves of E. obtusifolius were used to estimate the level of lipid peroxidation following a modified protocol of Tyagi et al. (2017). Briefly, leaf samples were homogenized in 2 mL 0.1% trichloroacetic acid and centrifuged at 11 000 × g for 10 min. Thereafter, 1 mL 0.25% thiobarbituric acid in 10% trichloroacetic acid was added to 1 mL supernatant. The reaction mixture was heated at 95 °C in a water bath for 30 min and then immediately cooled on ice. The absorbance of the reaction mixture was measured at 532 and 600 nm. MDA content was calculated using the extinction coefficient of MDA (ε = 155 mM−1 cm−1) and expressed as nmol g−1 FW.
Histochemical Detection of Hydrogen Peroxide (H2O2)
Endogenously produced H2O2 was visualized using the 3′3-diaminobenzidine (DAB) staining protocol described by Yang et al. (2013). Leaf samples of E. obtusifolius were submerged in 1 mg/mL DAB solution at room temperature, pH 3.8 for 8 h under illumination. After 8 h, the leaves were boiled in 95% (v/v) ethanol for 10 min to decolourize the leaves (remove pigments) except for the deep brown polymerization product of the DAB reaction with H2O2. After cooling, the bleached leaves were preserved in 4:1 (v/v) ethanol-glycerol solution and kept at 4 ºC until photographed. Intense brown colouration indicated a higher concentration of H2O2 in the leaves.
Phytochemical Content
Total phenolic and flavonoid content in the leaves of E. obtusifolius were measured following the colourimetric methods described by Dragović-Uzelac et al. (2007). Briefly, ethyl acetate extracts were prepared from leaf samples. Total phenolic content was evaluated using Folin-Ciocalteu reagent. The optical density was measured at 725 nm and the total phenolic content expressed as mg g−1 gallic acid equivalents. Flavonoid content was evaluated using aluminium chloride where the optical density was measured at 510 nm. Flavonoid content was expressed as mg g−1 catechin equivalents.
1-1-Diphenyl-1-picrylhydrazyl (DPPH) Radical Scavenging Activity
The radical scavenging activity of the leaves was measured following the method of Sridhar and Charles (2019). Briefly, crude ethyl acetate extracts were prepared from leaf samples and mixed with 0.1 mM DPPH solution prepared in methanol. Following 30 min incubation in the dark, the optical density was measured at 517 nm. The IC50 value of each extract was calculated using a non-linear regression curve of the % inhibition (scavenging activity) against the logarithm of concentrations. Butylated hydroxytoluene (BHT) was included as a standard.
Ferric-reducing Antioxidant Power (FRAP) Assay
The iron reducing power was measured following the method of Moyo et al. (2013). Ethyl acetate leaf extracts were serially diluted in microwell plates and a 1% (w/v) potassium ferricyanide reaction mixture added. Following incubation for 30 min at 50 °C, the mixture was acidified with trichloroacetic acid (1% in phosphate buffer) and then distilled water and ferric chloride (0.1% in phosphate buffer) added. Absorbance of the mixture was measured at 630 nm. OD values were plotted against their concentration gradient and slope values calculated. BHT was included as a standard.
α-Glucosidase Inhibitory Assay
The hypoglycemic activity of the E. obtusifolius leaves was measured as described by Rengasamy et al. (2013). Briefly, crude ethyl acetate leaf extracts were diluted in 5% dimethyl sulfoxide and 95% phosphate buffer (0.02 M, pH 6.9) containing 0.006 M NaCl to varying concentrations (10–100 µg mL−1). This was mixed with α-amylase enzyme solution (2 U mL−1) and incubated for 10 min at room temperature, after which 1% starch solution in phosphate buffer was added to initiate the reaction. The reaction was halted after 10 min by the addition of 3,5-dinitrosalicylic acid reagent and heating at 95 °C for 10 min. The optical density was measured at 540 nm. The enzyme inhibition percentage was calculated and IC50 values determined by extrapolation from the standard curve. The experiment was done in triplicate and acarbose included as a standard.
Statistical Analysis
The data was analyzed statistically based on the experimental design with a two-way analysis of variance using GraphPad Prism 7 (GraphPad Software, Inc. California USA). Mean comparison was done with the Bonferroni post hoc test at a 5% significance level (p ≤ 0.05).
Results
Establishment of Inoculation Colonies
Endophytic species colonizing the roots of E. obtusifolius were isolated from the inoculated root segments. They were identified as P. polymyxa and F. oxysporum based on the morphological characters of the initial cultures. This indicated that the method was effective for the successful inoculation of E. obtusifolius stem cuttings with the two isolated endophytes.
Growth of E. obtusifolius
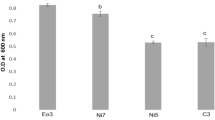
The main factors (watering regime and endophyte inoculation) and their synergistic effects significantly influenced the root and shoot biomass of E. obtusifolius plants (Table 1). The F-values and P-values were used to assess the level of significance of the factors and their interaction (Table 1). Probability (P) values less than 0.05 were considered significant. Control plants subjected to mild water stress (50% FC) had significantly higher shoot and root dry weights compared to the other watering regimes (Figs. 1 and 2). All endophyte inoculation treatments significantly increased growth of E. obtusifolius with the highest biomass accumulation in the well-watered (100% FC), endophyte-treated plants (Figs. 1 and 2). F. oxysporum and co-inoculation applications elicited the best response in the shoots (Fig. 2a) and roots (Fig. 2b) of well watered and mild water stressed plants. Under severe water stress, the co-inoculation treatments were the most effective in increasing shoot growth (Fig. 2a). Co-inoculation treatments elicited the highest root DW across the three watering regimes.
Growth of Endostemon obtusifolius stem cuttings subjected to various water stress levels for 8 weeks when inoculated with two endophytes. WW = well-watered (100% FC), MS = mild stress (50% FC), SS = severe stress (25% FC), a = uninoculated control, b = inoculated with Paenibacillus polymyxa, c = inoculated with Fusarium oxysporum and d = co-inoculated with P. polymyxa + F. oxysporum
a Shoot and b root dry weight of Endostemon obtusifolius stem cuttings subjected to various water stress levels for 8 weeks when inoculated with two endophytes. Results are presented as mean ± standard error (n = 3) where different letter(s) indicate a significant difference (p < 0.05) between treatments
Photosynthetic Pigments and Chlorophyll Fluorescence
The concentration of photosynthetic pigments in E. obtusifolius leaves were significantly affected by water stress, individual inoculation and co-inoculation with P. polymyxa and F. oxysporum (Table 1). The total chlorophyll content in E. obtusifolius increased significantly as the severity of water stress increased. Inoculation treatments had a positive effect with significantly higher chlorophyll content in the F. oxysporum (well-watered) and co-inoculation (mild and severe stressed) treated plants (Fig. 3a). Mild water stressed plants had a significantly higher carotenoid content compared to the well watered and severely water stressed plants. The carotenoid content decreased in the mild water stressed plants when treated with the inoculums. Inoculation, especially with P. polymyxa significantly increased the carotenoid content in well watered and severely water stressed plants (Fig. 3b).
a Chlorophyll and b carotenoid content and c chlorophyll fluorescence in leaves from Endostemon obtusifolius stem cuttings subjected to various water stress levels for 8 weeks when inoculated with two endophytes. Results are presented as mean ± standard error (n = 3) where different letter(s) indicate a significant difference (p < 0.05) between treatments
Photochemical efficiency as measured by chlorophyll fluorescence (FV/FM) was significantly affected by water stress and inoculation treatments (Table 1). There was no difference between the FV/FM values of uninoculated plants under well watered and mild stress conditions but values decreased significantly in plants grown under severe water stress (Fig. 3c). Single inoculation treatments only increased chlorophyll fluorescence in the severe water stressed plants while the co-inoculation treatment (P + FI) increased chlorophyll fluorescence under all watering regimes (Fig. 3c).
RWC and Electrolyte Leakage
The severity of water stress, endophyte inoculation and their interactive effects significantly affected the RWC of E. obtusifolius leaves (Table 1). The RWC decreased in both untreated and inoculated E. obtusifolius plants as the severity of water stress increased (Fig. 4a). F. oxysporum inoculation was the most effective in improving the RWC of E. obtusifolius under well watered conditions and the co-inoculation treatment was the most effective under severe stress conditions. Endophyte inoculation treatments had no effect on the RWC on plants subjected to mild water stress (Fig. 4a).
a Relative water content and b electrolyte leakage in leaves of Endostemon obtusifolius stem cuttings subjected to various water stress levels for 8 weeks when inoculated with two endophytes. Results are presented as mean ± standard error (n = 3) where different letters indicate a significant difference (p < 0.05) between treatments
The electrolyte leakage of E. obtusifolius was influenced by the individual factors although their synergistic effects were not statistically significant (Table 1). In general, there was an increase in electrolyte leakage as drought stress became more severe with the highest electrolyte leakage in the uninoculated plants under severe stress conditions (Fig. 4b). F. oxysporum and co-inoculation treatments significantly reduced electrolyte leakage with the lowest value in the well watered plants (Fig. 4b).
Osmolytes (Total Soluble Sugar and Proline Content)
Water stress, endophyte inoculation and their interactive effects significantly affected the total soluble sugar and proline contents in leaves of E. obtusifolius (Table 1). Uninoculated plants subjected to mild water stress had the highest total soluble sugar (Fig. 5a) and proline (Fig. 5b) content compared to well watered and severely stressed uninoculated plants. Generally, endophyte inoculation significantly promoted soluble sugar and proline accumulation across the three watering regimes with the co-inoculation treatment consistently being the most effective (Fig. 5a and b).
a Total soluble sugar and b proline content in leaves of Endostemon obtusifolius stem cuttings subjected to various water stress levels for 8 weeks when inoculated with two endophytes. Results are presented as mean ± standard error (n = 3) where different letters indicate a significant difference (p < 0.05) between treatments
Antioxidant Systems (SOD Activity, MDA Content and H2O2 Visualization)
Drought stress, endophyte inoculation and their interactive effects caused significant changes to SOD activity in E. obtusifolius (Table 1). Uninoculated plants subjected to mild water stress had higher SOD activity compared to uninoculated plants under well watered and severe stress conditions (Fig. 6a). Endophyte inoculation significantly increased SOD activity in plants grown under well watered (F. oxysporum inoculation) and severe stress conditions (co-inoculation treatment) but had no effect on plants grown under mild water stress (Fig. 6a).
Changes in a superoxide dismutase activity and b malondialdehyde content in Endostemon obtusifolius subjected to various water stress levels when inoculated with two endophytes. Results are presented as mean ± standard error (n = 3) where different letters indicate a significant difference (p < 0.05) between treatments
While drought stress caused significant changes in the MDA content in E. obtusifolius, endophyte inoculation had no effect (Table 1). MDA content increased significantly in mild and severe water stressed plants compared to well watered plants (Fig. 6b). Endophyte inoculation reduced lipid peroxidation in mild and severe water stressed plants although the lower MDA content was not significantly different in the uninoculated plants. Endophyte inoculation had no effect on the MDA content in well watered plants (Fig. 6b).
Histochemical visualization of H2O2 accumulation in the leaves of E. obtusifolius showed that DAB polymerization products increased under water stress (Fig. 7). Uninoculated plants grown under well watered (100% FC) and severe water stress (25% FC) conditions had higher amounts of DAB product compared to inoculated plants, indicating a reduction in H2O2 accumulation in inoculated plants. There was little or no visual differences between inoculated and uninoculated plants grown under mild water stress (50% FC; Fig. 7).
Histochemical visualization of hydrogen peroxide accumulation in leaves of Endostemon obtusifolius stem cuttings grown under three watering regimes when inoculated with two endophytes. WW well-watered, MS mild stress, SS severe stress, a = uninoculated treatment, b = P. polymyxa inoculated, c = F. oxysporum inoculated and d = P. polymyxa and F. oxysporum co-inoculated
Phytochemicals (Total Phenolic and Flavonoid Content)
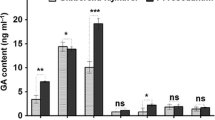
The total phenolic and flavonoid contents of E. obtusifolius were significantly altered by the watering regime, endophyte inoculation and their interactive effects (Table 1). Uninoculated plants subjected to mild water stress had higher total phenolic (Fig. 8a) and flavonoid (Fig. 8b) content compared to uninoculated plants under well watered and severe stress conditions. The phytochemical content in the mild water stressed plants was not significantly affected by single endophyte inoculation but significantly increased with co-inoculation (Fig. 8). Both single and co-inoculation endophyte treatments significantly increased the phytochemical content in well watered and severe water stressed plants (Fig. 8).
Antioxidant Activity (DPPH and FRAP Assays)
The radical scavenging capabilities of E. obtusifolius as measured in the DPPH and FRAP assays were significantly affected by the watering regimes, endophyte inoculation and their interactive effects (Table 1). Antioxidant activity of ethyl acetate extracts of leaf samples in the uninoculated treatments was similar in the DPPH and FRAP assays regardless of the watering regime (Fig. 9a and b). Endophyte inoculation significantly improved the radical scavenging abilities measured in the DPPH assay with co-inoculation treatments eliciting the lowest IC50 values. Co-inoculated plants subjected to mild water stress had the most potent DPPH activity (Fig. 9a). Co-inoculation had less effect on antioxidant activity as measured in the FRAP assay. While endophyte inoculation had no effect in the mild water stressed plants, extracts made from endophyte inoculated plants were more active (higher slope values) in the well watered and severe water stress treatments (Fig. 9b).
a DPPH and b FRAP antioxidant activity and c α-glucosidase activity in Endostemon obtusifolius subjected to various water stress levels when inoculated with two endophytes. Activity of antioxidant butylated hydroxytoluene (a and b) and acarbose (c) standards are indicated. Results are presented as mean ± standard error (n = 3) where different letters indicate a significant difference (p < 0.05) between treatments
α-Glucosidase Inhibitory Activity
α-Glucosidase retardation activity of E. obtusifolius leaves was significantly altered by the water regimes, endophyte inoculation and their interactive effects (Table 1). The hypoglycemic properties of uninoculated E. obtusifolius plants significantly improved as water stress levels decreased (Fig. 9c). Endophyte inoculation significantly increased the inhibitory activity (lower IC50 values) in mild and severe water stressed plants. F. oxysporum and co-inoculated treatments were the most effective. Endophyte inoculation treatments had no effect in the well watered plants (Fig. 9c).
Discussion
The root tissues of E. obtusifolius used in the present study were successfully colonized by the bacterium P. polymyxa and fungus F. oxysporum when applied to the soil surrounding the plantlets. The attachment and subsequent colonization of plants by endophytes depend on their rhizospheric presence, their motility rate towards the prospective host and their ability to break down plant cell walls (Mengistu 2020). The endophyte strains used in the present experiment were previously isolated from E. obtusifolius (Ogbe et al. 2023b) and were thus predisposed towards colonizing the plantlets established for this study.
As E. obtusifolius occurs naturally in arid and semi-arid regions, it is adapted to grow under water deficit conditions. In the present study, uninoculated plantlets subjected to mild water stress had significantly higher growth compared to well watered and severe water stressed uninoculated plants. Individual and co-inoculations with P. polymyxa and F. oxysporum significantly improved plant growth under all watering regimes (Figs. 1 and 2). Similarly, growth was improved with microbial inoculations in some indigenous plants such as Allium cepa ( Gana et al. 2022), Glycyrrhiza uralensis (Xie et al. 2019), Astragalus mongholicus (Sun et al. 2019) and Piper nigrum (Anith et al. 2018). Endophytes generally promote plant growth under normal and stressful conditions by improving the uptake of nutrients and water and through the regulation of growth and stress phytohormones (Afzal et al. 2019). Previously, 26 endophytes were isolated from E. obtusifolius and screened for growth-promoting characteristics. The bacteria P. polymyxa and fungus F. oxysporum isolates were selected as potential inoculi as they had good drought resistance capabilities combined with a number of beneficial plant-growth promoting characteristics. These included ammonia production, sidophore production, hydrogen cyanide, phosphate solubilization with P. polymyxa also having indole-3-acetic acid production and aminocyclopropane-1-carboxylate deaminase activity (Ogbe et al. 2023b).
Drought stress affects chlorophyll content differently in plants depending on plant tolerance and the severity of the stress factor. Generally, drought stress reduces photosynthetic pigments in plants ( Qi and Yin 2023) but in E. obtusifolius, the total chlorophyll content significantly increased with drought stress severity (Fig. 3a). Similarly, drought stress induced higher chlorophyll concentrations in Solanum tuberosum (Ramírez et al. 2014), clones of Eucalyptus grandii × E. robusta (Michelozzi et al. 1995) and Nicotiana tobacum (Gubiš et al. 2007). Endophyte inoculation significantly increased the chlorophyll content in E. obtusifolius (Fig. 3a) as in Helianthus tuberosus (Suebrasri et al. 2020) and Citrus reticulata (Sadeghi et al. 2020). Alteration in chlorophyll degradation during routine senescence occasioned by the “stay-green effect” may be responsible for the higher chlorophyll content in plants under drought stress (Ramírez et al. 2014). This can lead to a rise in ROS accumulation as the chlorophyll continues to trap and transfer energy onto oxygen molecules (Hörtensteiner 2009). Carotenoid content was also enhanced with endophyte inoculation in well watered and severely water stressed E. obtusifolius plantlets (Fig. 3b). Similarly, carotenoid content increased in endophyte-treated C. reticulata (Sadeghi et al. 2020). Carotenoids may function as antioxidant protection to the photosynthetic system to mitigate the damage caused by excessive ROS accumulation (Al-Arjani et al. 2020).
Chlorophyll fluorescence is used as a stress marker where Fv/Fm ratio values less than 0.7 imply that the plant is not tolerant to prevailing conditions (Bu et al. 2012). Endostemon obtusifolius plantlets subjected to severe drought stress treatment had a decline in Fv/Fm ratios below 0.7 (Fig. 3c), indicating photoinhibition due to protein damage and subsequent disruptions in the photochemical activities at Photosystem II (PSII) reaction centers (Banks 2017). Endophyte treatments (individual and co-inoculation) improved the photochemical efficiency of PSII in both drought stressed and unstressed E. obtusifolius plants with the highest Fv/Fm ratio in the co-inoculated, mild water stressed plants (Fig. 3c). Endophytes had a positive influence on the maximum quantum yield in Stevia rebaudiana (Saravi et al. 2021) and C. reticulata (Sadeghi et al. 2020). Endophytes improve the photochemical efficiency of PSII via the stabilization of grana structure, synthesis of protective metabolites and the enhancement of nitrogen and potassium absorption (Al-Kahtani et al. 2020).
The leaf RWC decreased in E. obtusifolius grown under severe drought stress, however, it was significantly improved with endophyte treatments (Fig. 4a). RWC reflects the current physiological status of the plant and can be linked to enhanced water and nutrient transportation, stomatal regulation and efficient hydraulic conductivity in plants (Zhang et al. 2019). Endophyte inoculation led to a significant reduction in electrolyte leakage in E. obtusifolius grown under the three watering regimes (Fig. 4b). Drought stress would elicit higher accumulation of ROS, causing lipid peroxidation and a deterioration to cellular membrane integrity, thus leading to a rise in electrolyte leakage (Emami-Bistgani et al. 2017). Endophyte inoculation mitigated the effects of excessive ROS generation in drought stressed E. obtusifolius as indicated by lower oxidative stress markers such as MDA (Fig. 6b) and H2O2 (Fig. 7).
Endophyte inoculation influenced an array of stress tolerance mechanisms which resulted in improved drought tolerance in E. obtusifolius. This included enhanced accumulation of osmolytes such as soluble sugar and proline (Fig. 5). The accumulation of such metabolites under water shortage is a typical stress response (Dastogeer et al. 2018; Al-Arjani et al. 2020). Soluble sugars help maintain turgor pressure under drought stress (Emami-Bistgani et al. 2017). The amino acid proline assists in maintaining the integrity and fluidity of cellular and molecular structures, photosynthetic apparatus (Abd-Allah et al. 2015), osmotic homeostasis (Al-Arjani et al. 2020) and scavenges free radicals (Chiappero et al. 2019). The enzymatic antioxidant system of E. obtusifolius was also enhanced with endophyte treatments where there was an increased SOD activity (Fig. 6a). Similarly, SOD activity was up-regulated in drought stressed Bacillus pumilus inoculated G. uralensis (Xie et al. 2019; Zhang et al. 2019). SOD is an antioxidant enzyme that converts O2− to hydrogen peroxide, which is further reduced to water and oxygen by the activities of other enzymatic and non-enzymatic antioxidants (Das and Roychoudhury 2014). Concentrations of antioxidant metabolites such as total phenolics and flavonoids were also increased in E. obtusifolius treated with endophyte inoculation (Fig. 8). The increase in these metabolites is an additional layer of defense to mitigate against the negative consequences of drought stress (Bettaieb et al. 2011). Other indigenous plants inoculated with microbial endophytes also have a higher phenolic and flavonoid content (Mona et al. 2017). Thus the endophyte inoculations used in the present study altered metabolite content and influenced some physiological processes in E. obtusifolius, thereby improving its growth and stress tolerance.
These physiological changes elicited by the endophytes resulted in increased antioxidant activity in leaf extracts of E. obtusifolius. Mild water stressed plantlets treated with co-inoculum had the highest antioxidant capacity in the DPPH assay. This activity was significantly better than the BHT standard (Fig. 9a). Endophyte inoculation significantly enhanced the antioxidant capacity of well watered plantlets in the FRAP assay to levels similar to the BHT standard (Fig. 9b). Similarly, antioxidant capacities of Stevia rebaudiana and L. citriodora increased with endophyte inoculation (Kilam et al. 2015; Golparyan et al. 2018). Application of these beneficial endophytes also enhanced the accumulation of other bioactive metabolites in E. obtusifolius where the in vitro α-glucosidase inhibitory potential of E. obtusifolius improved significantly in water stressed plants (Fig. 9c). Similarly, drought stress improved the antidiabetic activities of Hibiscus sabdariffa (Ifie et al. 2018) and endophytic bacteria inoculation was effective in enhancing antidiabetic activity in Momordica charantia (Pujiyanto and Ferniah 2017).
In conclusion, although E. obtusifolius grows in semi-arid and arid conditions, drought stress negatively affected the growth (root and shoot biomass), carotenoid content, chlorophyll fluorescence and relative water content. Under increasing water deficit conditions, E. obtusifolius generated more ROS which caused peroxidation of cellular molecules such as lipids and negatively affected cellular membrane integrity. Individual and co-inoculation treatments with P. polymyxa and F. oxysporum mitigated excessive ROS generation via increased osmolyte production (soluble sugars and proline), up-regulation of the enzymatic antioxidant system (SOD) and increased antioxidant metabolite content (total phenolics and flavonoids). This suggests that E. obtusifolius has established a symbiotic relationship with these beneficial endophytes to enhance its stress mechanisms under drought stress conditions. The pharmacological potential (antioxidant and in vitro α-glucosidase inhibitory activities) of E. obtusifolius leaves was significantly improved with endophyte inoculation with the most active extracts made from leaves of mild water stressed plants. This study highlighted a potential strategy in the cultivation of medicinal plants where drought stress combined with beneficial endophyte isolates can be used to enhance the productivity and efficacy of the crop.
Data Availability
The data is available upon request from the corresponding author (JvS).
References
Abd-Allah EF, Hashem A, Alqarawi AA, Bahkali AH, Alwhibi MS (2015) Enhancing growth performance and systemic acquired resistance of medicinal plant Sesbania sesban (L.) Merr using arbuscular mycorrhizal fungi under salt stress. Saudi J Biol Sci 22:274–283. https://doi.org/10.1016/j.sjbs.2015.03.004
Afzal I, Shinwari ZK, Sikandar S, Shahzad S (2019) Plant beneficial endophytic bacteria: mechanisms, diversity, host range and genetic determinants. Microbiol Res 221:36–49. https://doi.org/10.1016/j.micres.2019.02.001
Al-Arjani ABF, Hashem A, Abd Allah EF (2020) Arbuscular mycorrhizal fungi modulate dynamics tolerance expression to mitigate drought stress in Ephedra foliata Boiss. Saudi J Biol Sci 27:380–394
Al-Kahtani MDF, Attia KA, Hafez YM, Khan N, Eid AM, Ali MAM, Abdelaal KAA (2020) Chlorophyll fluorescence parameters and antioxidant defense system can display salt tolerance of salt acclimated sweet pepper plants treated with chitosan and plant growth promoting rhizobacteria. Agronomy 10:1180. https://doi.org/10.3390/agronomy10081180
Alamgir ANM (2017) Cultivation of herbal Drugs, biotechnology, and in vitro production of secondary metabolites, high-value medicinal plants, herbal wealth, and herbal trade. Therapeutic use of medicinal plants and their extracts, vol 1. Springer, Berlin, pp 379–452. https://doi.org/10.1007/978-3-319-63862-1_9
Anith KN, Aswini S, Varkey S, Radhakrishnan NV, Nair DS (2018) Root colonization by the endophytic fungus Piriformospora indica improves growth, yield and piperine content in black pepper (Piper nigurm L). Biocat Agric Biotechnol 14:215–220
Banks JM (2017) Continuous excitation chlorophyll fluorescence parameters: a review for practitioners. Tree Physiol 37:1128–1136. https://doi.org/10.1093/treephys/tpx059
Bettaieb I, Hamrouni-Sellami I, Bourgou S, Limam F, Marzouk B (2011) Drought effects on polyphenol composition and antioxidant activities in aerial parts of Salvia officinalis L. Acta Physiol Plant 33:1103–1111. https://doi.org/10.1007/s11738-0100638-z
Bibi N, Jan G, Jan FG, Hamayun M, Iqbal A, Hussain A, Rehman H, Tawab A, Khushdil F (2019) Cochliobolus sp. acts as a biochemical modulator to alleviate salinity stress in Okra plants. Plant Physiol Biochem 139:459–469. https://doi.org/10.1016/j.plaphy.2019.04.019
Bistgani ZE, Siadat SA, Bakhshandeh A, Ghasemi Pirbalouti A, Hashemi M (2017) Interactive effects of drought stress and chitosan application on physiological characteristics and essential oil yield of Thymus daenensis Celak. Crop J 5:407–415. https://doi.org/10.1016/j.cj.2017.04.003
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Bu N, Li X, Li Y, Ma C, Ma L, Zhang C (2012) Effects of Na2CO3 stress on photosynthesis and antioxidative enzymes in endophyte infected and non-infected rice. Ecotoxicol Environ Saf 78:35–40. https://doi.org/10.1016/j.ecoenv.2011.11.007
Caser M, Chitarra W, D’Angiolillo F, Perrone I, Demasi S, Lovisolo C, Pistelli L, Pistelli L, Scariot V (2019) Drought stress adaptation modulates plant secondary metabolite production in Salvia dolomitica Codd. Ind Crops Prod 129:85–96. https://doi.org/10.1016/j.indcrop.2018.11.068
Chiappero J, Cappellari L, del Sosa Alderete R, Palermo LG, Banchio TB E (2019) Plant growth promoting rhizobacteria improve the antioxidant status in Mentha piperita grown under drought stress leading to an enhancement of plant growth and total phenolic content. Ind Crops Prod 139:111553. https://doi.org/10.1016/j.indcrop.2019.111533
Das K, Roychoudhury A (2014) Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front Environ Sci 2:53. https://doi.org/10.3389/fenvs.2014.00053
Das P, Sarkar B, Mandal S (2023) Endophytic microbes: a potential source of bioactive metabolites with therapeutic values. In: Shah M, Deka D (eds) Endophytic association: what, why and how. Academic Press, Cambridge, pp 435–457. https://doi.org/10.1016/B978-0-323-91245-7.00004-3
Dastogeer KMG, Li H, Sivasithamparam K, Jones MGK, Wylie SJ (2018) Fungal endophytes and a virus confer drought tolerance to Nicotiana benthamiana plants through modulating osmolytes, antioxidant enzymes and expression of host drought responsive genes. Environ Exp Bot 149:95–108. https://doi.org/10.1016/j.envexpbot.2018.02
Dragović-Uzelac V, Levaj B, Bursać D, Pedisić S, Radojčić I, Biško A (2007) Total phenolics and antioxidant capacity assays of selected fruits. Agric Conspec Sci 72:279–284. https://hrcak.srce.hr/19384
Gana LP, Etsassala NG, Nchu F (2022) Interactive effects of water deficiency and endophytic Beauveria bassiana on plant growth, nutrient uptake, secondary metabolite contents, and antioxidant activity of Allium cepa L. J Fungi 8:874. https://doi.org/10.3390/jof8080874
Golparyan F, Azizi A, Soltani J (2018) Endophytes of Lippia citriodora enhance its growth and antioxidant activity. Eur J Plant Path 152:759–768. https://doi.org/10.1007/s10658-018-1520-x
Gubiš J, Vaňková R, Červená V, Dragúňová M, Hudcovicová M, Lichtnerová H, Dokupil T, Jureková Z (2007) Transformed tobacco plants with increased tolerance to drought. S Afr J Bot 73:505–511. https://doi.org/10.1016/j.sajb.2007.03.011
Hoagland DR, Snyder WC (1933) Nutrition of strawberry plants under controlled conditions. Proc Am Soc Hortic Sci 30:288–296
Hörtensteiner S (2009) Stay-green regulates chlorophyll and chlorophyll-binding protein degradation during senescence. Trends Plant Sci 14:155–162. https://doi.org/10.1016/j.tplants.2009.01.002
Hussain Z, Arslan M, Malik MH, Mohsin M, Iqbal S, Afzal M (2018) Treatment of the textile industry effluent in a pilot-scale vertical flow constructed wetland system augmented with bacterial endophytes. Sci Total Environ 645:966–973. https://doi.org/10.1016/j.scitotenv.2018.07.163
Ifie I, Ifie BE, Ibitoye DO, Marshall LJ, Williamson G (2018) Seasonal variation in Hibiscus sabdariffa (Roselle) calyx phytochemical profile, soluble solids, and α-glucosidase inhibition. Food Chem 261:164–168. https://doi.org/10.1016/j.foodchem.2018.04.052
Kakar N, Jumaa SH, Sah SK, Redoña ED, Warburton ML, Reddy KR (2022) Genetic variability assessment of tropical indica rice (Oryza sativa L.) seedlings for drought stress tolerance. Plants 11:2332. https://doi.org/10.3390/plants11182332
Kapoor D, Bhardwaj S, Landi M, Sharma A, Ramakrishnan M, Sharma A (2020) The impact of drought in plant metabolism: how to exploit tolerance mechanisms to increase crop production. Appl Sci 10:5692. https://doi.org/10.3390/app10165692
Khan Z, Rho H, Firrincieli A, Han Hung S, Luna V, Masciarelli O, Kim S-H, Doty SL (2016) Growth enhancement and drought tolerance of hybrid poplar upon inoculation with endophyte consortia. Curr Plant Biol 6:38–47. https://doi.org/10.1016/j.cpb.2016.08.001
Khan A, Pan X, Najeeb U, Tan DKY, Fahad S, Zahoor R, Luo H (2018) Coping with drought: stress and adaptive mechanisms, and management through cultural and molecular alternatives in cotton as vital constituents for plant stress resilience and fitness. Biol Res 51:1–17. https://doi.org/10.1186/540659-018-019-z
Khare E, Mishra J, Arora NK (2018) Multifaceted interactions between endophytes and plant: developments and prospects. Front Microbiol 9:2732. https://doi.org/10.3389/fmicb.2018.02732
Kilam D, Saifi M, Abdin MZ, Agnihotri A, Varma A (2015) Combined effects of Piriformospora indica and Azotobacter chroococcum enhance plant growth, antioxidant potential and steviol glycoside content in Stevia rebaudiana. Symbiosis 66:149–156. https://doi.org/10.1007/s13199-015-0347-x
Kumar M, Sharma S, Gupta S, Kumar V (2018) Mitigation of abiotic stresses in Lycopersicon esculentum by endophytic bacteria. Environ Dev Sustain 1:71–80. https://doi.org/10.1007/s42398-018-0004-4
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Meth Enzymol 148:350–382. https://doi.org/10.1016/0076-6879(87)48036-1
Lu H, Wei T, Lou H, Shu X, Chen Q (2021) A critical review on communication mechanism within plant-endophytic fungi interactions to cope with biotic and abiotic stresses. J Fungi 7:719. https://doi.org/10.3390/jof7090719
Mengistu AA (2020) Endophytes: colonization, behaviour, and their role in defense mechanism. Int J Microbiol 2020:6927219. https://doi.org/10.1155/2020/6927219
Michelozzi M, Johnson JD, Warrag EI (1995) Response of ethylene and chlorophyll in two Eucalyptus clones during drought. New for 9:197–204. https://doi.org/10.1007/BF00035487
Mona SA, Hashem A, Abd Allah EF, Alqarawi AA, Soliman DWK, Wirth S, Egamberdieva D (2017) Increased resistance of drought by Trichoderma harzianum fungal treatment correlates with increased secondary metabolites and proline content. J Integr Agric 16:1751–1757. https://doi.org/10.1016/S2095-3119(17)61695-2
Moyo M, Amoo SO, Ncube B, Ndhlala AR, Finnie JF, van Staden J (2013) Phytochemical and antioxidant properties of unconventional leafy vegetables consumed in southern Africa. S Afr J Bot 84:65–71. https://doi.org/10.1016/j.sajb.2012.09.010
Nwafor I, Nwafor C, Manduna I (2021) Constraints to cultivation of medicinal plants by smallholder farmers in South Africa. Hortic 7:531. https://doi.org/10.3390/horticulturae7120531
Ogbe AA, Gupta S, Finnie JF, van Staden J (2023a) Preliminary in vitro antioxidant and retardation of essential carbohydrate hydrolysing enzymes by some indigenous South African medicinal plants. S Afr J Bot 159:686–696. https://doi.org/10.1016/j.sajb.2023.05.030
Ogbe AA, Gupta S, Stirk WA, Finnie JF, van Staden J (2023b) Growth-promoting characteristics of fungal and bacterial endophyes isolated from a drought-tolerant mint species Endostemon obtusifolius (E. Mey. ex Benth) N. E. Br. Plants 12:638. https://doi.org/10.3390/plants12030638
Ong WY, Wu YJ, Farooqui T, Farooqui AA (2018) Qi Fu Yin—a Ming dynasty prescription for the treatment of dementia. Mol Neurobiol 55:7389–7400. https://doi.org/10.1007/s12035-018-0908-0
Ouyang Y, Cheng Q, Cheng C, Tang Z, Huang Y, Tan E, Ma S, Lin X, Xie Y, Zhou H (2023) Effects of plants-associated microbiota on cultivation and quality of Chinese herbal medicines. Chin Herb Med. https://doi.org/10.1016/j.chmed.2022.12.004
Pujiyanto S, Ferniah RS (2017) Alpha-glucosidase inhibitor activity of Momordica charantia L. after inoculated by endophytic bacteria. Adv Sci Lett 23:6490–6492. https://doi.org/10.1166/asl.2017.9662
Qi J, Yin D (2023) Effects of Suillus luteus on the growth, photosynthesis, stomata, and root system of Pinus tabulaeformis under drought stress. J Plant Growth Regul 42:3486–3497. https://doi.org/10.1007/s00344-022-10809-9
Ramakuwela T, Hatting J, Bock C, Vega FE, Wells L, Mbata GN, Shapiro-Ilan D (2020) Establishment of Beauveria bassiana as a fungal endophyte in pecan (Carya illinoinensis) seedlings and its virulence against pecan insect pests. Biol Control 140:104102. https://doi.org/10.1016/j.biocontrol.2019.104102
Ramírez DA, Yactayo W, Gutiérrez R, Mares V, De Mendiburu F, Posadas A, Quiroz R (2014) Chlorophyll concentration in leaves is an indicator of potato tuber yield in water-shortage conditions. Sci Hortic 168:202–209.
Rengasamy KRR, Aderogba MA, Amoo SO, Stirk WA, van Staden J (2013) Potential antiradical and alpha-glucosidase inhibitors from Ecklonia maxima (Osbeck) Papenfuss. Food Chem 14:1412–1415. https://doi.org/10.1016/j.foodchem.2013.04.019
Rho H, Hsieh M, Kandel SL, Cantillo J, Doty SL, Kim SH (2018) Do endophytes promote growth of host plants under stress? A meta-analysis on plant stress mitigation by endophytes. Microb Eco 75:407–418. https://doi.org/10.1007/s00248-01-1054-3
Sadashiva CT, Naidoo Y, Naidoo JR, Kalicharan B, Naidoo G (2014) Antioxidant and acetylcholinesterase activities of three species of the family Lamiaceae. Bangladesh J Bot 43:331–335. https://doi.org/10.3329/bjb.v43i3.21606
Sadeghi F, Samsampour D, Askari Seyahooei M, Bagheri A, Soltani J (2020) Fungal endophytes alleviate drought-induced oxidative stress in mandarin (Citrus reticulata L.): toward regulating the ascorbate–glutathione cycle. Sci Hortic 261:1–11. https://doi.org/10.1016/j.scienta.2019.108991
Salvi P, Mahawar H, Agarrwal R, Gautam V, Deshmukh R (2022) Advancement in the molecular perspective of plant-endophytic interaction to mitigate drought stress in plants. Front Microbiol 13:981355. https://doi.org/10.3389/fmicb.2022.981355
Saravi A, Gholami H, Pirdashti MB, Firouzabadi HA (2021) The response of Stevia (Stevia rebaudiana Bertoni) photosystem II photochemistry to fungi symbiosis and spermidine application under saline water irrigation. Russ Agric Sci 47:32–36. https://doi.org/10.3101/S106836742101016X
Seile BP, Bareetseng S, Koitsiwe MT, Aremu AO (2022) Indigenous knowledge on the uses, sustainability and conservation of African ginger (Siphonochilus aethiopicus) among two communities in Mpumalanga Province, South Africa. Diversity 14:192. https://doi.org/10.3390/d14030192
Sridhar K, Charles AL (2019) In vitro antioxidant activity of Kyoho grape extracts in DPPH (rad) and ABTS (rad) assays: estimation methods for EC50 using advanced statistical programs. Food Chem 275:41–49. https://doi.org/10.1016/j.foodchem.2018.09.040
Suebrasri T, Harada H, Jogloy S, Ekprasert J, Boonlue S (2020) Auxin-producing fungal endophytes promote growth of sunchoke. Rhizosphere 16:100271. https://doi.org/10.1016/j.rhisph.2020.100271
Sun H, Kong L, Du H, Chai Z, Gao J, Cao Q (2019) Benefits of Pseudomonas poae s61 on Astragalus mongholicus growth and bioactive compound accumulation under drought stress. J Plant Interact 14:205–212. https://doi.org/10.1080/17429145.2019.1611958
Tamokou JDD, Mbaveng AT, Kuete V (2017) Antimicrobial activities of African medicinal spices and vegetables. In: Kuete V (ed) Medicinal spices and vegetables from Africa. Academic Press, Cambridge, pp 207–237. https://doi.org/10.1016/B978-0-12-809286-6.00008.X
Tyagi J, Varma A, Pudake RN (2017) Evaluation of comparative effects of arbuscular mycorrhiza (Rhizophagus intraradices) and endophyte (Piriformospora indica) association with finger millet (Eleusine coracana) under drought stress. Eur J Soil Biol 81:1–10. https://doi.org/10.1016/j.ejsobi.2017.05.007
Xie Z, Chu Y, Zhang W, Lang D, Zhang X (2019) Bacillus pumilus alleviates drought stress and increases metabolite accumulation in Glycyrrhiza uralensis Fisch. Environ Exp Bot 158:99–106. https://doi.org/10.1016/j,envexpbot.2018.11.021
Yang W, Zhu C, Ma X, Li G, Gan L, Ng D, Xia K (2013) Hydrogen peroxide is a second messenger in the salicylic acid-triggered adventitious rooting process in mung bean seedlings. PLoS ONE 8:1–14. https:/doi.org/10.137/journal.pone.0084580
Yang HR, Yuan J, Liu LH, Zhang W, Chen F, Dai CC (2019) Endophytic Pseudomonas fluorescens induced sesquiterpenoid accumulation mediated by gibberellic acid and jasmonic acid in Atractylodes macrocephala Koidz plantlets. Plant Cell Tissue Organ Cult 138:445–457. https://doi.org/10.1007/s11240-019-01640-4
Ye HT, Luo SQ, Yang ZN, Wang YS, Ding Q, Wang KF, Yang SX, Wang Y (2021) Endophytic fungi stimulate the concentration of medicinal secondary metabolites in Houttuynia cordata Thunb. Plant Signal Behav 16:1–11. https://doi.org/10.1080/15592325.2021.1929731
Yue L, Uwaremwe C, Tian Y, Liu Y, Zhao X, Zhou Q, Wang Y, Zhang Y, Liu B, Cui Z, Dun C (2022) Bacillus amyloliquefaciens rescues glycyrrhizic acid loss under drought stress in Glycyrrhiza uralensis by activating the jasmonic acid pathway. Front Microbiol 12:798525. https://doi.org/10.3389/fmicb.2021.798525
Zhang W, Xie Z, Zhang X, Lang D, Zhang X (2019) Growth-promoting bacteria alleviates drought stress of Glycyrrhiza uralensis through improving photosynthesis characteristics and water status. J Plant Interact 14:580–589. https://doi.org/10.1080/17429145.2019.1680752
Acknowledgements
Mrs Alison Young is thanked for providing technical assistance in raising the stem cuttings.
Funding
Open access funding provided by University of KwaZulu-Natal. This work was supported by the National Research Foundation, South Africa (Grant No. 145740 and Grant No. CSRP2204041882) and the University of KwaZulu-Natal.
Author information
Authors and Affiliations
Contributions
AAO, SG and JvS conceptualized the study. The experiments and data collection was carried out by AAO and SG. AAO performed the data analysis. AAO wrote the first draft and WAS prepared the figures and reviewed and edited the manuscript. AAO was supervised by JvS and JFF. Resources and funding were provided by JvS and SG. All authors read and approved the final manuscript. WAS is an editor of this journal but was not involved in the evaluation of this manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interests.
Additional information
Handling Editor: Peter Hedden.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ogbe, A.A., Gupta, S., Stirk, W.A. et al. Endophyte Inoculation Enhances Growth, Secondary Metabolites and Biological Activity of Endostemon obtusifolius Grown Under Drought Stress. J Plant Growth Regul 43, 1103–1117 (2024). https://doi.org/10.1007/s00344-023-11167-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-023-11167-w