Abstract
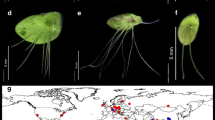
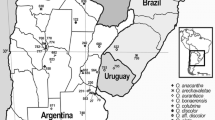
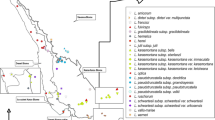
Amplified fragment length polymorphism (AFLP) fingerprinting and three different plastidic DNA regions (rpl16, rps16, atpF-atpH) were used to investigate species identity in the genus Wolffiella. For this purpose, clones (67 in total) belonging to all ten species were selected. Almost all the species were represented by more than one clone. The fingerprinting technique, AFLP, clearly distinguished the species, W. caudata, W. gladiata, W. neotropica, W. rotunda, and W. welwitschii. Apart from confirming the molecular identity of these five species, the plastidic markers could delineate two additional species, W. hyalina and W. denticulata, although the conclusion concerning the latter is restricted by the availability of only one clone. The efficiency of the plastid-derived markers in identifying the number of species-specific clusters followed the sequence rps16 > rpl16 > atpF-atpH. The species W. lingulata, W. oblonga, and W. repanda could not be identified by any of the molecular methods presented here, but could be strictly defined on a morphological basis. In several clones, high amounts of genetic admixtures between different species were detected. Further, simulation studies demonstrated that these clones are genetic hybrids. This might be one of the obstacles in molecular identification of species in the genus Wolffiella.






Similar content being viewed by others
References
Appenroth K-J, Teller S, Horn M (1996) Photophysiology of turion formation and germination in Spirodela polyrhiza. Biol Pl 38:95–106. https://doi.org/10.1007/BF02879642
Appenroth KJ, Borisjuk N, Lam E (2013) Telling duckweed apart: genotyping technologies for Lemnaceae. Chin J Appl Environm Biol 19:1–10. https://doi.org/10.3724/SP.J.1145.2013.00001
Appenroth KJ, Crawford DJ, Les DH (2015) After the genome sequencing of duckweed—how to proceed with research on the fastest growing angiosperm? Pl Biol (Stuttgart) 17(Suppl 1):1–4. https://doi.org/10.1111/plb.12248
Balao F, García-Castaño JL (2014) AFLPsim: an R package to simulate and detect dominant markers under selection in hybridizing populations. Pl Meth 10:40. https://doi.org/10.1186/1746-4811-10-40
Baumbach H, Hellwig FH (2007) Genetic differentiation of metallicolous and non-metallicolous Armeria maritima (Mill.) Willd. taxa (Plumbaginaceae) in Central Europe. Pl Syst Evol 269:245–258. https://doi.org/10.1007/s00606-007-0593-3
Bog M, Baumbach H, Schween U, Hellwig F, Landolt E, Appenroth K-J (2010) Genetic structure of the genus Lemna L. (Lemnaceae) as revealed by amplified fragment length polymorphism. Planta 232:609–619. https://doi.org/10.1007/s00425-010-1201-2
Bog M, Schneider P, Hellwig F, Sachse S, Kochieva EZ, Martyrosian E, Landolt E, Appenroth K-J (2013) Genetic characterization and barcoding of taxa in the genus Wolffia Horkel ex Schleid. (Lemnaceae) as revealed by two plastidic markers and amplified fragment length polymorphism (AFLP). Planta 237:1–13. https://doi.org/10.1007/s00425-012-1777-9
Bog M, Lautenschlager U, Landrock MF, Landolt E, Fuchs J, Sree KS, Oberprieler C, Appenroth KJ (2015) Genetic characterization and barcoding of taxa in the genera Landoltia and Spirodela (Lemnaceae) by three plastidic markers and amplified fragment length polymorphism (AFLP). Hydrobiologia 749:169–182. https://doi.org/10.1007/s10750-014-2163-3
Borisjuk N, Chu P, Gutierrez R, Zhang H, Acosta K, Friesen N, Sree KS, Garcia C, Appenroth KJ, Lam E (2015) Assessment, validation and deployment strategy of a two barcode protocol for facile genotyping of duckweed species. Pl Biol (Stuttgart) 17(Suppl 1):42–49. https://doi.org/10.1111/plb.12229
Coughlan NE, Kelly TC, Jansen MAK (2017) Step by step: high frequency short-distance epizoochorous dispersal of aquatic macrophytes. Biol Invasions 19:625–634. https://doi.org/10.1007/s10530-016-1293-0
Crawford DJ, Landolt E, Les DH, Tepe E (1997) Allozyme variation and the taxonomy of Wolffiella. Aquatic Bot 58:43–54. https://doi.org/10.1016/S0304-3770(97)00012-0
Doyle JJ, Dickson EE (1987) Preservation of plant samples for DNA restriction endonuclease analysis. Taxon 36:715–722. https://doi.org/10.2307/1221122
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Falush D, Stephens M, Pritchard JK (2007) Inference of population structure using multilocus genotype data: dominant markers and null alleles. Molec Ecol Notes 7:574–578. https://doi.org/10.1111/j.1471-8286.2007.01758.x
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acidc Symp 41:95–98
Hegelmaier F (1895) Systematische Uebersicht der Lemnaceen. Bot Jahrb 21:268–305
Huson DH, Bryant D (2006) Application of phylogenetic networks in evolutionary studies. Molec Biol Evol 23:254–267. https://doi.org/10.1093/molbev/msj030
International Steering Committee on Duckweed Research and Applications (2013) [Recommendations for registration of duckweed clones by the International Steering Committee on Duckweed Research and Applications]. ISCDRA Newsletter 1(1):3–4. Available at: http://www.ruduckweed.org. Accessed 24 June 2017
Jordan WC, Courtney MW, Neigel JE (1996) Low levels of intraspecific genetic variation at a rapid evolving chloroplast DNA locus in North American duckweeds (Lemnaceae). Amer J Bot 83:430–439. https://doi.org/10.2307/2446212
Kimball RT, Crawford DJ, Les DH, Landolt E (2003) Out of Africa: molecular phylogenetics and biogeography of Wolffiella (Lemnaceae). Biol J Linn Soc 79:565–576. https://doi.org/10.1046/j.1095-8312.2003.00210.x
Landolt E (1984) Flowers and fruits in the genus Wolffiella (Lemnaceae). Ber Geobot Inst ETH Stiftung Rübel 51:164–172
Landolt E (1986) The family of Lemnaceae—a monographic study. Vol. 1. Biosystematic Investigations in the Family of Duckweeds (Lemnaceae), vol 1. Veröffentlichungen des Geobotanischen Institutes der ETH, Stiftung Ruebel, Zürich
Landolt E (1992) Wolffiella caudata, a new Lemnaceae species from the Bolivian Amazon region. Ber Geobot Inst ETH Stiftung Rübel 58:121–123
Les DH, Crawford DJ, Landolt E, Gabel JD, Kimball RT (2002) Phylogeny and systematics of Lemnaceae, the duckweed family. Sys Bot 27:221–240
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452. https://doi.org/10.1093/bioinformatics/btp187
Meier R, Kwong S, Vaidya G, Ng PKL (2006) DNA barcoding and taxonomy in Diptera: a tale of high intraspecific variability and low identification success. Syst Biol 55:715–728. https://doi.org/10.1080/10635150600969864
Monod T (1949) Sur une Lemnacee africaine: Wolffiella welwitschii (Hegelmaier 1865) comb. nov. Mém Soc Hist Nat Afrique N, Hors Sér 2:229–242
Nei M, Li WH (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci USA 76:5269–5273. https://doi.org/10.1073/pnas.76.10.5269
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Wagner H (2016) vegan: Community Ecology Package. R package version 2.3-5. Available at: https://CRAN.R-project.org/package=vegan. Accessed 5 Jan 2017
Oxelman B, Liden M, Berglund D (1997) Chloroplast rps16 intron phylogeny of the tribe Silenae (Caryophyllaceae). Pl Sys Evol 206:393–410. https://doi.org/10.1007/BF00987959
Pieterse AH, Bhalla PR, Sabharwal PS (1971) Endogenous gibberellins in floating plants and turions of Wolffiella floridana. Physiol Pl 24:512–516. https://doi.org/10.1111/j.1399-3054.1971.tb03527.x
Pritchard JK (2010) Documentation for Structure software: Version 2.3. Available at: http://pritch.bsd.uchicago.edu/structure.html. Accessed 5 Jan 2017
Pritchard JK, Stephens M, Donelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Ryzhova NN, Martyrosian LV, Kolganova TV, Goryunova SV, Kochieva EZ (2006) Characterization of the trnL-trnF spacer sequence of the chloroplast tRNA genes in Spirodela species. Molec Biol 40:891–896. https://doi.org/10.1134/S0026893306060070
Small RL, Lickey EB, Shaw J, Hauk WD (2005) Amplification of noncoding chloroplast DNA for phylogenetic studies in lycophytes and monilophytes with a comparative example of relative phylogenetic utility from Ophioglossaceae. Molec Phylogen Evol 36:509–522. https://doi.org/10.1016/j.ympev.2005.04.018
Sree KS, Sudakaran S, Appenroth K-J (2015) How fast can angiosperms grow? species and clonal diversity of growth rates in the genus Wolffia (Lemnaceae). Acta Physiol Pl 37:204. https://doi.org/10.1007/s11738-015-1951-3
Sree KS, Bog M, Appenroth KJ (2016) Taxonomy of duckweeds (Lemnaceae), potential new crop plants. Emirate J Food Agricult 28:291–302. https://doi.org/10.9755/ejfa.2016-01-038
Swofford DL (2003) PAUP*—phylogenetic analysis using parsimony (*and other methods). Sinauer Associates, Sunderland
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molec Biol Evol 28:2731–2739. https://doi.org/10.1093/molbev/msr121
R Core Team (2016) R: A language and Environment for statistical computing. R Foundation for Statistical Computing, Vienna. Available at: https://www.R-project.org/. Accessed 05 Jan 2017
Tippery NP, Les DH, Crawford DJ (2015) Evaluation of phylogenetic relationships in Lemnaceae using nuclear ribosomal data. Pl Biol (Stuttgart) 17(Suppl 1):50–58. https://doi.org/10.1111/plb.12203
Vos P, Hogers R, Bleeker M, Reijans M, Van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M, Zabenau M (1995) AFLP: a new technique for DNA fingerprinting. Nucl Acids Res 23:4407–4414. https://doi.org/10.1093/nar/23.21.4407
Wang W, Wu Y, Yan Y, Ermakova M, Kerstetter R, Messing J (2010) DNA barcoding of the Lemnaceae, a family of aquatic monocots. BMC Pl Biol 10:205–214. https://doi.org/10.1186/1471-2229-10-205
Zhao H, Appenroth K, Landesman L, Salmean AA, Lam E (2012) Duckweed rising at Chengdu: summary of the 1st international conference on Duckweed application and research. Pl Molec Biol 78:627–632. https://doi.org/10.1007/s11103-012-9889-y
Ziegler P, Adelmann K, Zimmer S, Schmidt C, Appenroth K-J (2015) Relative in vitro growth rates of duckweeds (Lemnaceae)—the most rapidly growing higher plants. Pl Biol (Stuttgart) 17(Suppl 1):33–41. https://doi.org/10.1111/plb.12184
Acknowledgements
We thank the late Elias Landolt (1926–2013) for his essential contribution at the beginning of the present project and for providing majority of the clones. We also thank Ms Katja Kuehdorf for experimental support in the initial experiments. KSS is thankful to Science and Engineering Research Board, Government of India, Fast Track Young Scientist project for the support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Handling editor: Pablo Vargas.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Information on Electronic Supplementary Material
Information on Electronic Supplementary Material
Online Resource 1. 0/1 matrix of AFLP investigations of clones of Wolffiella.
Online Resource 2. Alignment of sequences related to Fig. 5a.
Online Resource 3. Alignment of sequences related to Fig. 5b.
Online Resource 4. Alignment of sequences related to Fig. 5c.
Online Resource 5. Alignment of sequences related to Fig. 6.
Online Resource 6. Cluster assignment of Wolffiella clones according to the ten independent runs from Structure for K = 9; run with highest lnPD highlighted in bold red.
Online Resource 7. Correlations between the maximum intraspecific distances and the minimum interspecific distances (K2P) for the regions rpl16, rps16, and atpF-atpH as well as all possible combinations.
Online Resource 8. Number of haplotypes per plastidic marker and for all possible combinations of the three markers for the sequenced Wolffiella clones. In addition, the species specificity of the haplotypes is indicated in an overview table.
Online Resource 9. Recalculated UPGMA tree based on atpF-atpH sequences from all 21 clones of Wolffiella investigated by Wang et al. (2010). In comparison with the published tree (Fig. 3 in Wang et al. 2010), also clones of W. denticulata 8221 and W. hyalina 8640 were included and the sequence of W. rotunda 9072 was revised.
Rights and permissions
About this article
Cite this article
Bog, M., Landrock, M.F., Drefahl, D. et al. Fingerprinting by amplified fragment length polymorphism (AFLP) and barcoding by three plastidic markers in the genus Wolffiella Hegelm. Plant Syst Evol 304, 373–386 (2018). https://doi.org/10.1007/s00606-017-1482-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-017-1482-z