Abstract
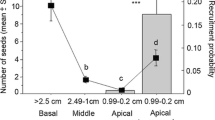
Mistletoes typically grow on tall old trees. Does this positive size-prevalence relationship result simply from the accumulation of infections as trees age, or do other factors related to tree size lead to differential dispersal, germination, establishment or survival of mistletoes? We examined patterns of infection prevalence and intensity of the mistletoe Phragmanthera dschallensis on its main host, Acacia sieberana, in a savannah in Zambia. The probability that an A. sieberana tree was infected increased with tree size, although infection intensity did not. In addition, infected trees were significantly taller than non-infected trees, even after removing the effect of tree age, using trunk diameter as a proxy. To determine whether differential dispersal occurred, we observed the feeding behaviour of the three main avian dispersers of this mistletoe, Pogoniulus chrysoconus, Lybius torquatus and Cinnyricinclus leucogaster, in relation to tree size. All three preferred perching in taller trees, whether trees were parasitized or not. To test whether differential germination of seeds or establishment of seedlings occurred, we planted mistletoe seeds on small and large A. sieberana trees and recorded germination, establishment and survival of seedlings. All seeds germinated, and seedling establishment at 7 months and seedling survival to 14 months was no different on small and large trees. Thus, host size did not affect germination and establishment. Finally, we measured the survival of established mistletoes on trees of different sizes over a 3-year period. Mistletoes survived significantly better on taller hosts. Our results support the hypotheses that higher mistletoe infection prevalence in taller trees results from differential dispersal of mistletoe seeds to tall trees as well as differential survival of established mistletoes on tall trees, and is not simply an accumulation of infections as trees age.




Similar content being viewed by others
References
Aukema J, Martínez Del Río C (2002a) Where does a fruit-eating bird deposit mistletoe seeds? Seed deposition patterns and an experiment. Ecology 83:3489–3496
Aukema J, Martínez Del Río C (2002b) Mistletoes as parasites and seed dispersing birds as disease vectors: current understanding, challenges and opportunities. In: Levey D, Silva W, Galetti M (eds), Seed dispersal and frugivory: ecology, evolution and conservation. CABI Publishing, Oxon, pp 99–110
Donohue K (1995) The spatial demography of mistletoe parasitism on a Yemeni Acacia. Int J Plant Sci 156:816–823
Fanshawe D (1969) The vegetation of Zambia. Division of Forestry Research, Kitwe
Godschalk S (1983) Mistletoe dispersal by birds in South Africa. In: Calder M, Bernhardt P (eds), The biology of mistletoes. Academic Press, San Diego, pp 117–128
Kelly P, Reid N, Davies I (1997) Effects of experimental burning, defoliation, and pruning on survival and vegetative resprouting in mistletoes (Amyema miquelii and Amyema pendula). Int J Plant Sci 158:856–861
Lamont B (1983) Germination of mistletoes. In: Calder M, Bernhardt P (eds), The biology of mistletoes. Academic Press, San Diego, pp 129–143
Lavorel S, Smith M, Reid N (1999) Spread of mistletoes (Amyema preissii) in fragmented Australian woodlands: a simulation study. Landscape Ecol 14:147–160
Maclean GL (1993) Roberts birds of southern Africa, 6th edn. John Voelcker Bird Book Fund, Cape Town, South Africa
Martínez Del Río C, Silva A, Medel R, Hourdequin M (1996) Seed dispersers as disease vectors: bird transmission of mistletoe seeds to plant hosts. Ecology 77:912–921
Medel R, Vergara E, Silva A, Kalin-Arroyo M (2004) Effects of vector behaviour and host resistance on mistletoe aggregation. Ecology 85:120–129
Niklas K (1995) Size-dependent allometry of tree height, diameter and trunk-taper. Ann Bot 75:217–227
Norton D, Ladley J, Owen H (1997) Distribution and population structure of the loranthaceous mistletoes Mepis flavida, Peraxilla colensoi, and Peraxilla tetrapetala within two New Zealand Nothofagus forests. N Z J Bot 35:323–336
Norton D, Reid N (1997) Lessons in ecosystem management from management of threatened and pest Loranthaceous mistletoes in New Zealand and Australia. Conserv Biol 11:759–769
Nowak DJ, McBride RR (1992) Differences in Moneterey pine pest populations in urban and natural forests. Forest Ecol Manage 50:133–144.
Overton J (1994) Dispersal and infection in mistletoe metapopulations. J Ecol 82:711–723
Polhill R, Wiens D (1998) Mistletoes of Africa. Royal Botanic Gardens, Kew, UK
Reid N (1991) Coevolution of mistletoes and frugivorous birds? Aust J Ecol 16:457–469
Reid N, Stafford Smith M (2000) Population dynamics of an arid zone mistletoe (Amyema preissii, Loranthaceae) and its host Acacia victoriae (Mimosaceae). Aust J Bot 48:45–58
Room P (1973) Ecology of the mistletoe Tapinanthus bangwensis growing on cocoa in Ghana. J Ecol 61:729–742
Ullmann I, Lange OL, Ziegler H, Ehleringer J, Schulze ED, Cowan IR (1985) Diurnal courses of leaf conductance and transpiration of mistletoes and their hosts in central Australia. Oecologia 67:577–587
Vermeulen S (1999) Distribution of mistletoes in a patchy habitat. PhD thesis, Imperial College, London
Yan Z (1993) Germination and seedling development of two mistletoes, Amyema preissii and Lysiana exocarpi: host specificity and mistletoe-host compatibility. Aust J Ecol 18:419–429
Acknowledgements
Thanks to Emma and Ian Bruce-Miller for their hospitality and for unlimited access to Nkanga River Conservation Area, and to Ruston Mukampola, our field assistant. Funding was provided by the University of Pretoria and the South African National Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Appendix 1. Host species of Phragmanthera dschallensis
Appendix 1. Host species of Phragmanthera dschallensis
-
1.
Acaciapolyacantha (Willd.) (Fabaceae)
-
2.
Acaciasieberana (DC.) (Fabaceae)
-
3.
Albiziaharveyi (Fourn.) (Fabaceae)
-
4.
Dichrostachyscinerea (L. Wight et Arn.) (Fabaceae)
-
5.
Faidherbiaalbida (Delile) A. Chev. (Fabaceae)
-
6.
Lonchocarpuscapassa (Rolfe) (Fabaceae)
-
7.
Combretumfragrans (F. Hoffm.) (Combretaceae)
-
8.
Combretummolle (R. Br. ex G. don.) (Combretaceae)
-
9.
Ficussycamorus (L.) (Moraceae)
-
10.
Ziziphusabyssinica (A. Rich.) (Rhamnaceae)
Rights and permissions
About this article
Cite this article
Roxburgh, L., Nicolson, S.W. Differential dispersal and survival of an African mistletoe: does host size matter?. Plant Ecol 195, 21–31 (2008). https://doi.org/10.1007/s11258-007-9295-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-007-9295-8