Quick filters:
Marattia Stock Photos and Images

RMD75N38–King Fern, Ptisana salicina (Syn. Marattia fraxinea, Marattia salicina) Marattiaceae. Australia and South Pacific.

RMDCFE8M–Marattia salicina, young whole plant, tropical glasshouse of the Jardin des Plantes of Paris

RM2HB32MC–Marattia. From the album: New Zealand ferns. 167 varieties, Eric Craig, maker/artist, 1888, Auckland, blueprint process

RF2HK6ND7–Inspired by Marattia. From the album: New Zealand ferns. 167 varieties, Eric Craig, maker/artist, 1888, Auckland, blueprint process, Reimagined by Artotop. Classic art reinvented with a modern twist. Design of warm cheerful glowing of brightness and light ray radiance. Photography inspired by surrealism and futurism, embracing dynamic energy of modern technology, movement, speed and revolutionize culture

RMPFJJRB–Ancient plants, including Cornus alba, Equisetum giganteum, Chamaecyparis, Cycas revoluta, Ginkgo biloba, Sequoia sempervirens, Prunus laurocerasus, Araucaria, Passiflora, Ilex aquifolium, Blechnum, Marattia werneri
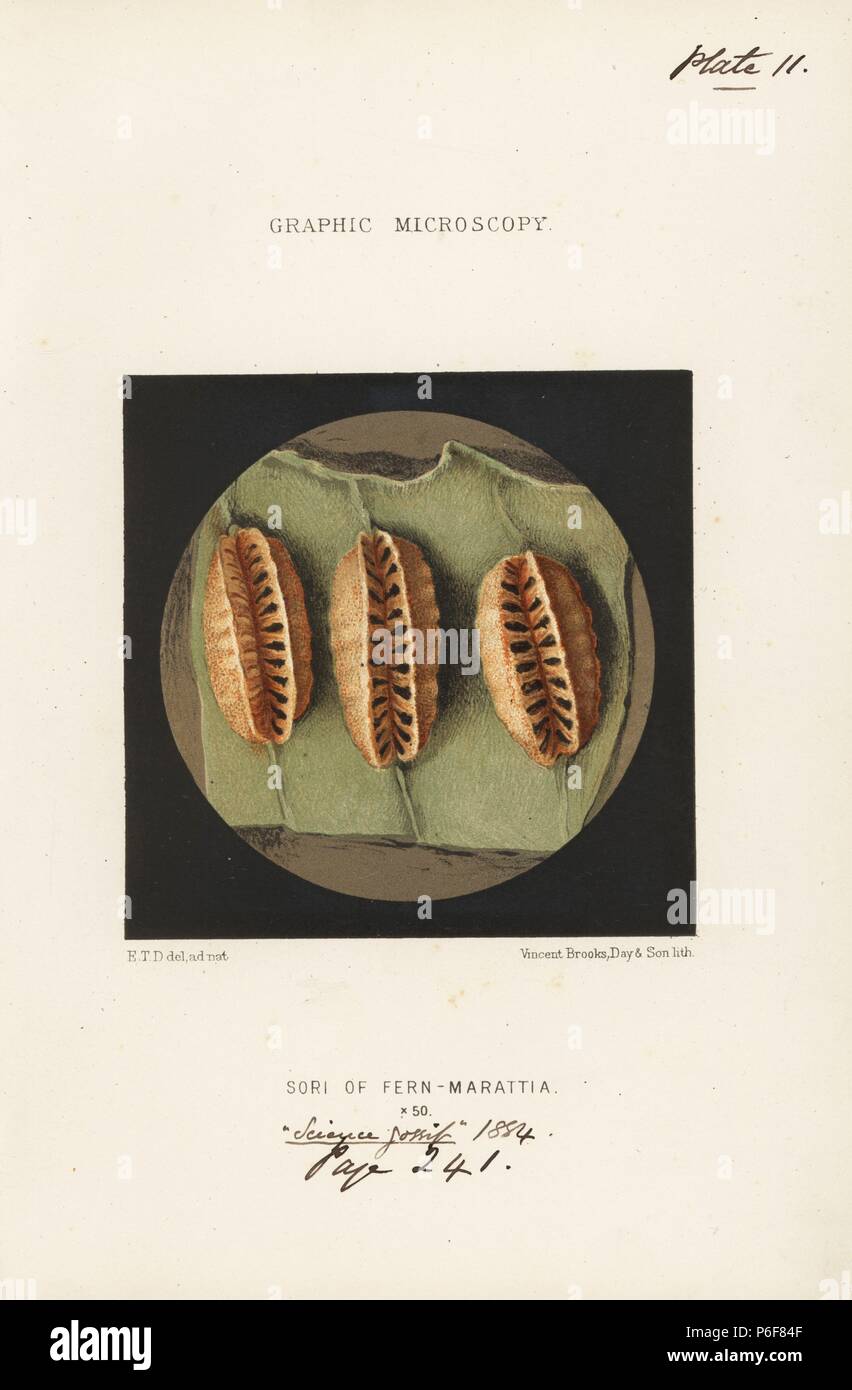
RMP6F84F–Sori or sporangia (spore cases) on the underside of a fern frond, Marattia alata, magnified x50. Chromolithograph after an illustration by E.T.D., lithographed by Vincent Brooks, from 'Graphic Microscopy' plates to illustrate 'Hardwicke's Science Gossip,' London, 1865-1885.
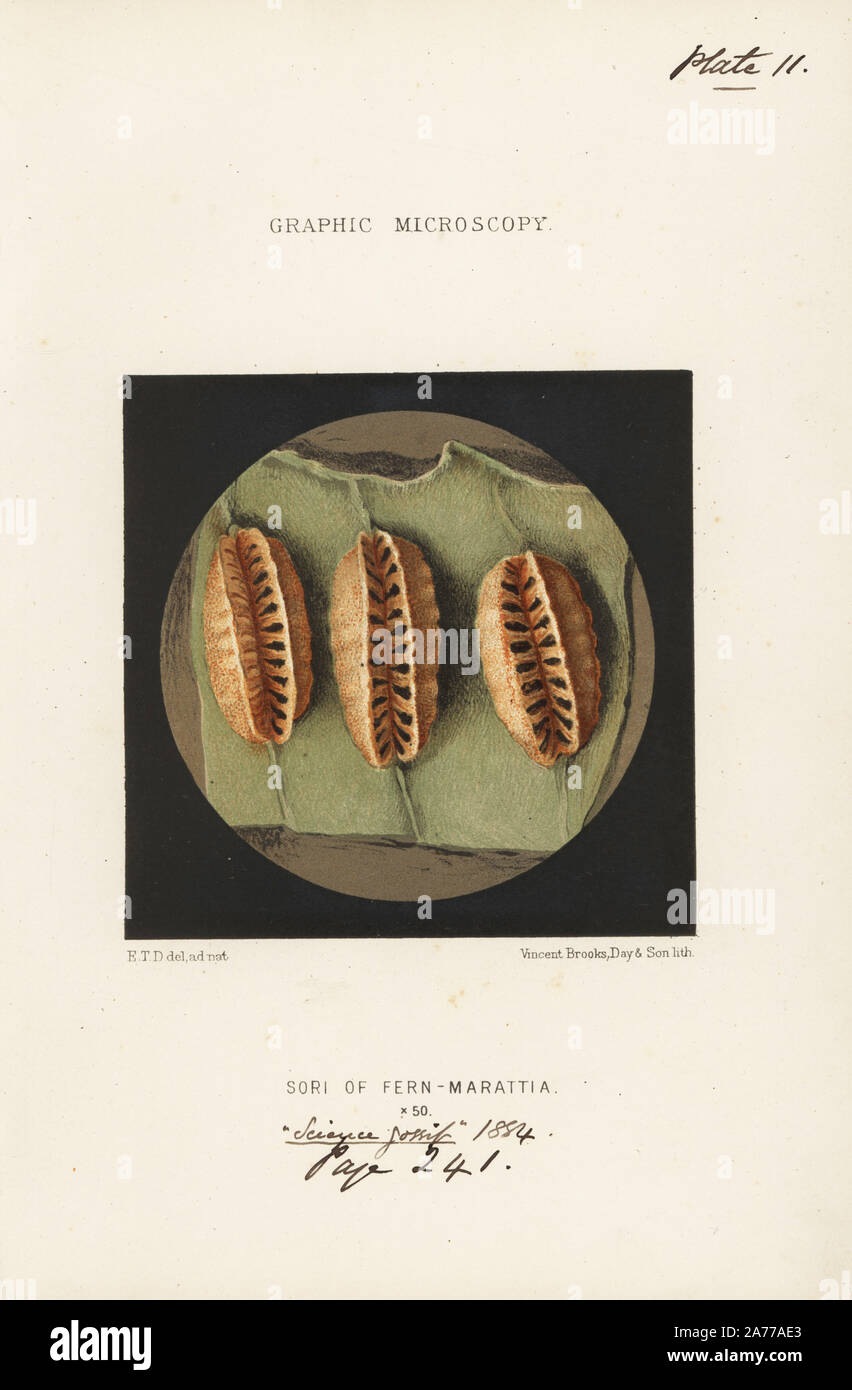
RM2A77AE3–Sori or sporangia (spore cases) on the underside of a fern frond, Marattia alata, magnified x50. Chromolithograph after an illustration by E.T.D., lithographed by Vincent Brooks, from 'Graphic Microscopy' plates to illustrate 'Hardwicke's Science Gossip,' London, 1865-1885.

RM2JRC4WN–Marattia fraxinia, Tuakau. From the album: New Zealand ferns,148 varieties, 1880, Auckland, by Herbert Dobbie.

RM2WTJRBN–Marattia fraxinia, Pokeno. From the album: New Zealand ferns. 172 varieties cyanotypes, photographic prints
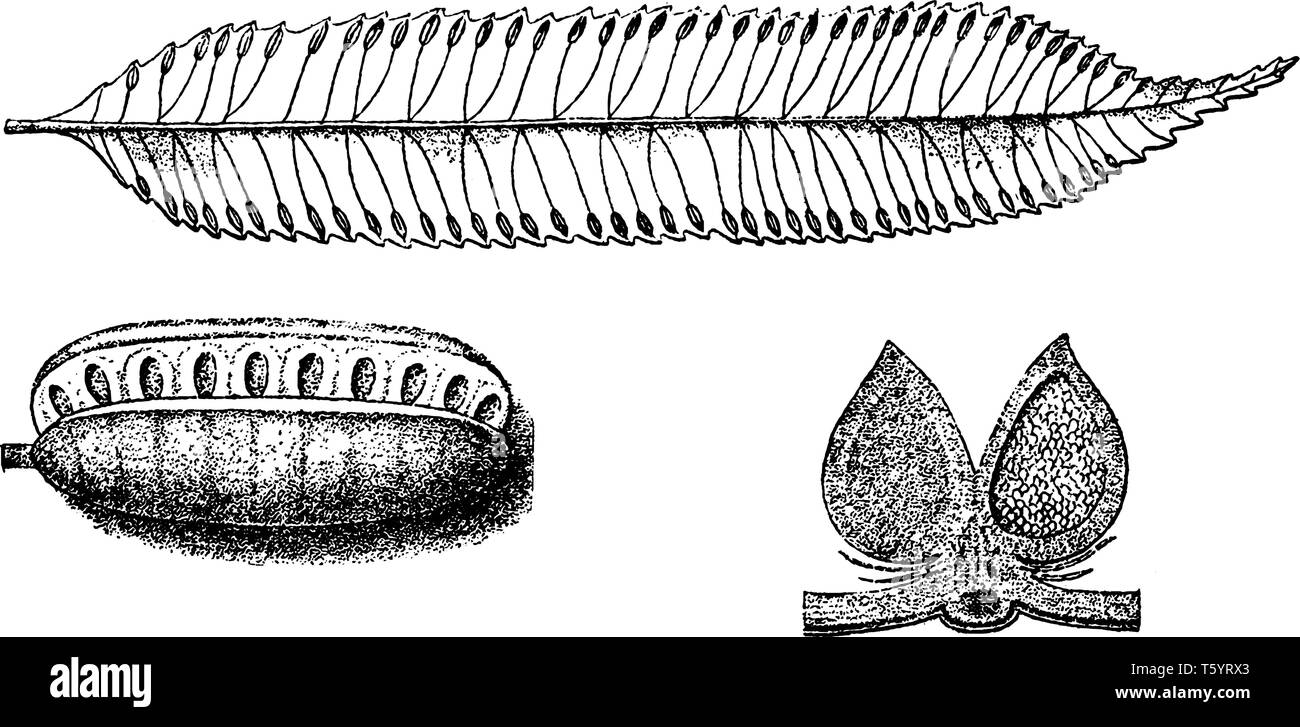
RFT5YRX3–Marattia is a genus of eusporangiate ferns. The plants are large and terrestrial, vintage line drawing or engraving illustration.

RM2A7C9KF–King fern, Ptisana salicina, native to Australia. Handcoloured copperplate stipple engraving from Jussieu's 'Dictionary of Natural Science,' Florence, Italy, 1837. Engraved by Stanghi, drawn by Pierre Jean-Francois Turpin, and published by Batelli e Figli. Turpin (1775-1840) is considered one of the greatest French botanical illustrators of the 19th century.
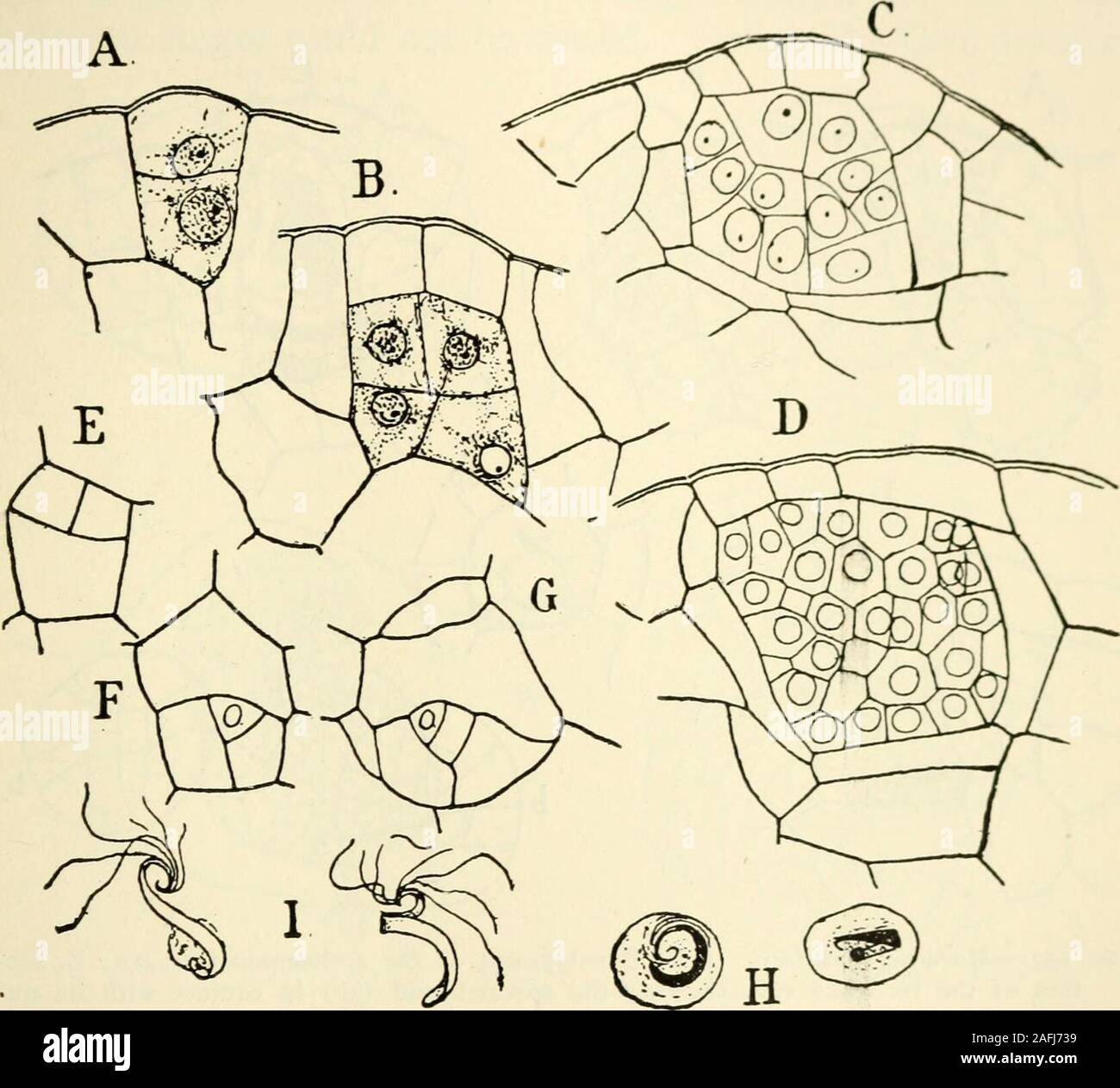
RM2AFJ739–. The structure and development of mosses and ferns (Archegoniatae). sperm cell, after its finaldivision, shows no nucleolus. The first sign of the formationof the spermatozoid that could be detected was an indentationupon one side, followed by a rapid flattening and growth of thewhole nucleus. The cytoplasmic prominence which, accordingto Strasburger, is the first indication of the formation of thespermatozoid, could not be certainly detected. The main partof the spermatozoid, stains strongly with alum-cochineal, andis sharply differentiated against the colourless cytoplasm, and VIII MARATTIA
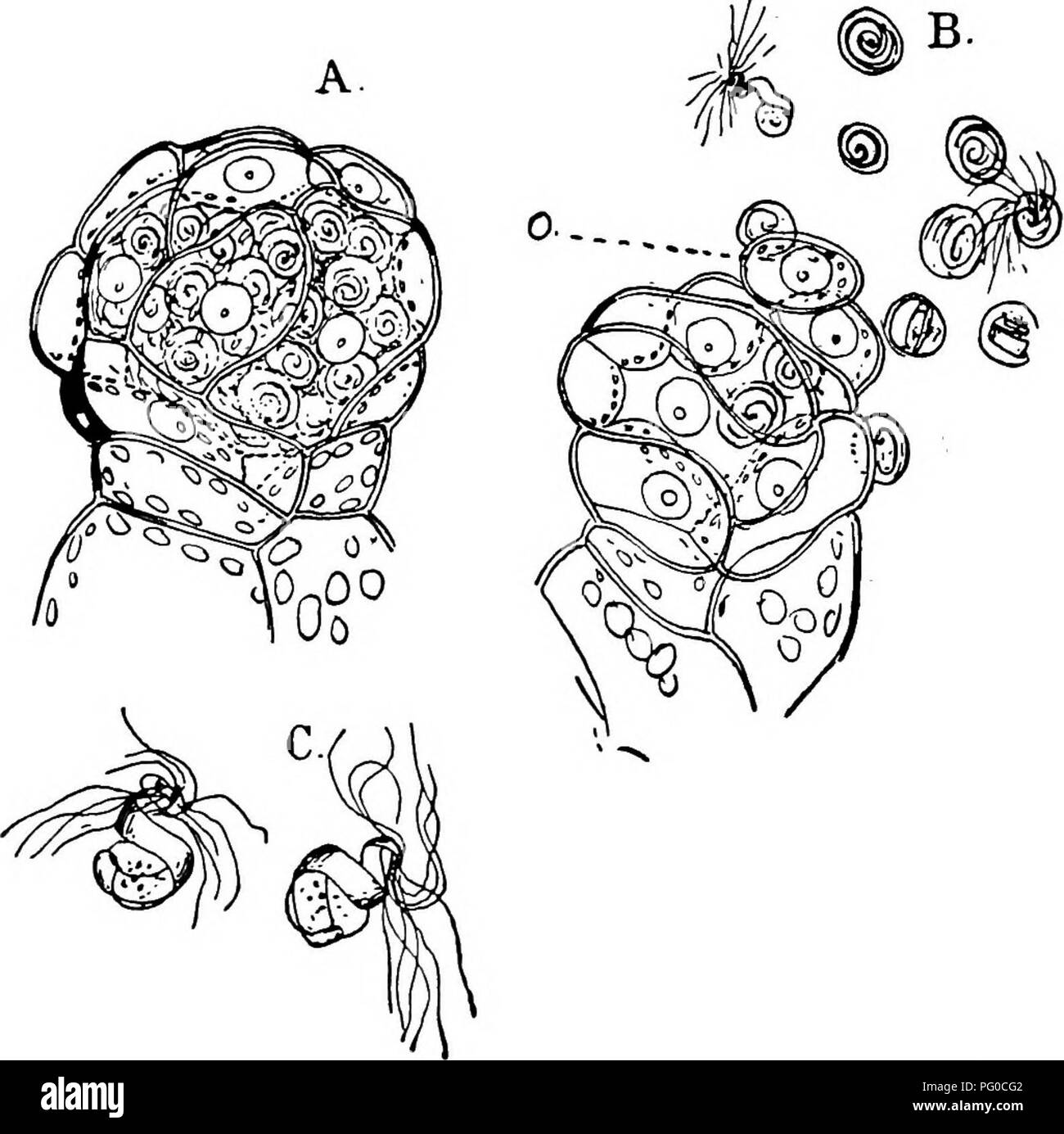
RMPG0CG2–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. THE HOMOSPOROUS LEPTOSPORANGIAT^ 353 as in the Hepaticse, and the mature spermatozoids are coiled more flatly than in the Polypodiacese. The free spermatozoid recalls that of Marattia or Equisetum rather than that of the Polypodiacese. There are but about two complete coils, and the hinder one relatively larger than in the latter forms. In swimming there is peculiar undulating movement, suggestive of the spermatozoid of Eqiiisctitvi. The Archegonium The archegonia are only borne upon the large

RMW075T0–Archive image from page 500 of Die farnkräuter der jetztwelt, zur. Die farnkräuter der jetztwelt, zur untersuchung und bestimmung, der in den formationen der erdrinde eingeschlossenen überreste von vorweltlichen arten dieser ordnung nach dem flächen-skelet diefarnkruterder00etti Year: 1865 Taf. 177. 3 4 5 1, 2. Marattia Kaulfnssii J. Smith. 3. Angiojiteris sp. (Mayotte). 4. Marattia Baddiana Schott. 5. 6. Psilodochea salicifolia P r e s 1.

RF2HKN3A5–Art inspired by Marattia. From the album: New Zealand ferns. 167 varieties, Eric Craig, maker/artist, 1888, Auckland, blueprint process, Classic works modernized by Artotop with a splash of modernity. Shapes, color and value, eye-catching visual impact on art. Emotions through freedom of artworks in a contemporary way. A timeless message pursuing a wildly creative new direction. Artists turning to the digital medium and creating the Artotop NFT

RMP6EEP5–King fern, Ptisana salicina, native to Australia. Handcoloured copperplate stipple engraving from Jussieu's 'Dictionary of Natural Science,' Florence, Italy, 1837. Engraved by Stanghi, drawn by Pierre Jean-Francois Turpin, and published by Batelli e Figli. Turpin (1775-1840) is considered one of the greatest French botanical illustrators of the 19th century.

RMMCRC1D–. B. Marattia fraxinea Smith. Im Schluchtenwald bei Araani in Oät-Usambara, um 900 m ü. d. M. Nach einer Photographie von Dr. Braun (1907).

RM2JRP76M–Marattia Fraxinia. From the album: New Zealand ferns. 148 varieties, 1880, Auckland, by Herbert Dobbie.
![Marattia fraxinia [fraxinea], Tuakau. From the album: New Zealand ferns. 148 varieties cyanotypes, photographic prints Stock Photo Marattia fraxinia [fraxinea], Tuakau. From the album: New Zealand ferns. 148 varieties cyanotypes, photographic prints Stock Photo](https://c8.alamy.com/comp/2WTKRY2/marattia-fraxinia-fraxinea-tuakau-from-the-album-new-zealand-ferns-148-varieties-cyanotypes-photographic-prints-2WTKRY2.jpg)
RM2WTKRY2–Marattia fraxinia [fraxinea], Tuakau. From the album: New Zealand ferns. 148 varieties cyanotypes, photographic prints

RM2JRPWFC–Marattia. From the album: New Zealand ferns. 167 varieties, 1888, Auckland, by Eric Craig, Herbert Dobbie.
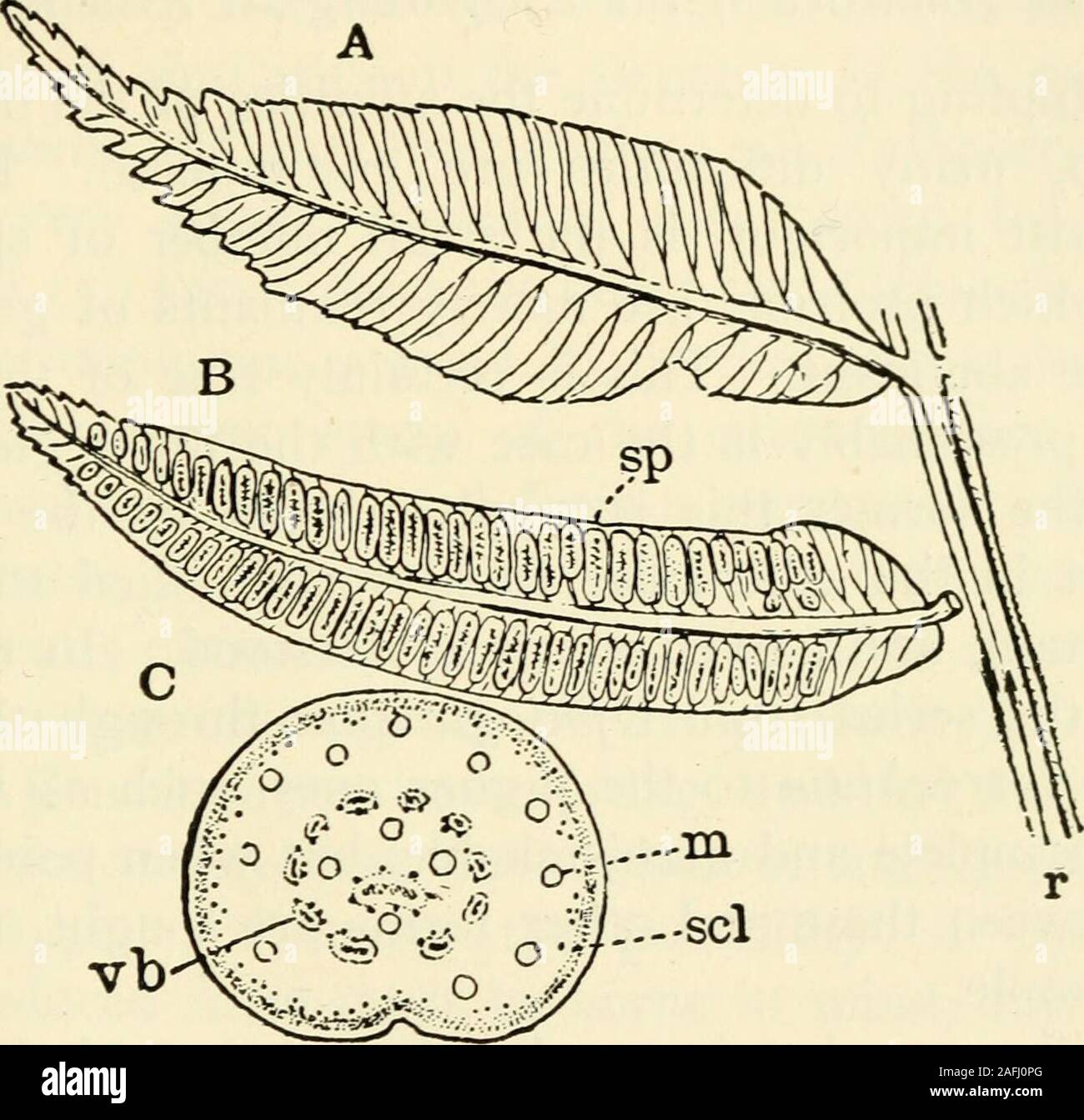
RM2AFJ0PG–. The structure and development of mosses and ferns (Archegoniatae). ication of the Marattiace^ - The living Marattiaceae (Bitter (i)) may be divided intofour sub-families, of which the first, Angiopteridese includestwo genera, Angiopteris and Archangiopteris, while the others,Marattiese, Kaulfussiese, and Danaease, contains each but asingle genus. - - VIII MARATTIALES 299 Marattia includes about twelve species of tropical and sub-tropical Ferns, both of the Old World and the New. Kaiil-fussia includes but a single species, belonging to southeasternAsia. The synangia are scattered over the low
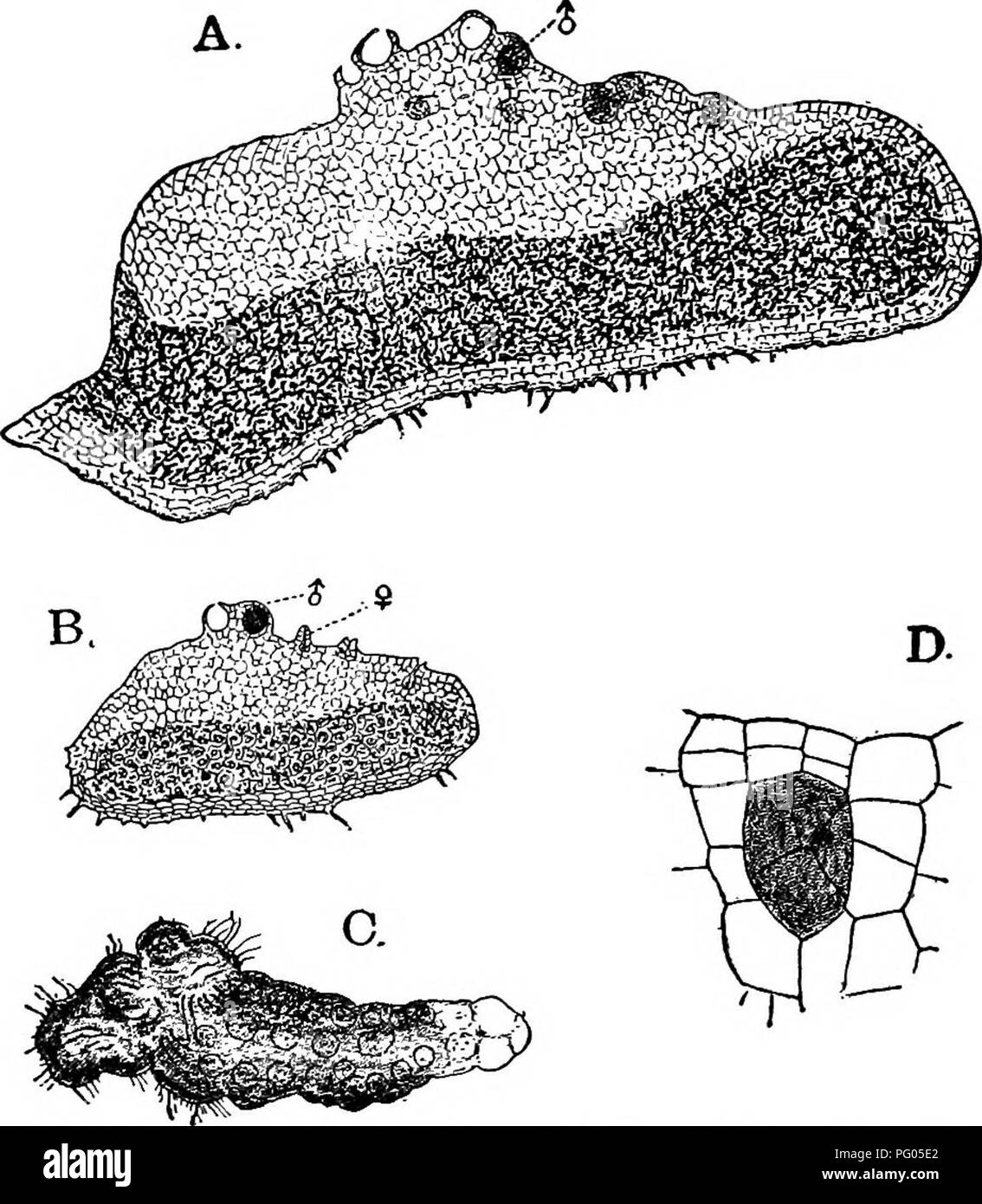
RMPG05E2–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. VII PTERIDOPHYTA—FILICINE^—OPHIOGLOSSACEJE 237 from which the cover of the antheridium is formed. The outer wall of the antheridium remains for the most part but one cell thick, in tljis respect more resembling Marattia than it does Botrychium. The antheridium also opens by a single, nearly triangular opercular cell (Fig. 125, E), as it does in Marattia. The spermatozoids were not seen, but probably resemble those of Botrychium or Marattia. The first division of the young archegonium is the sa
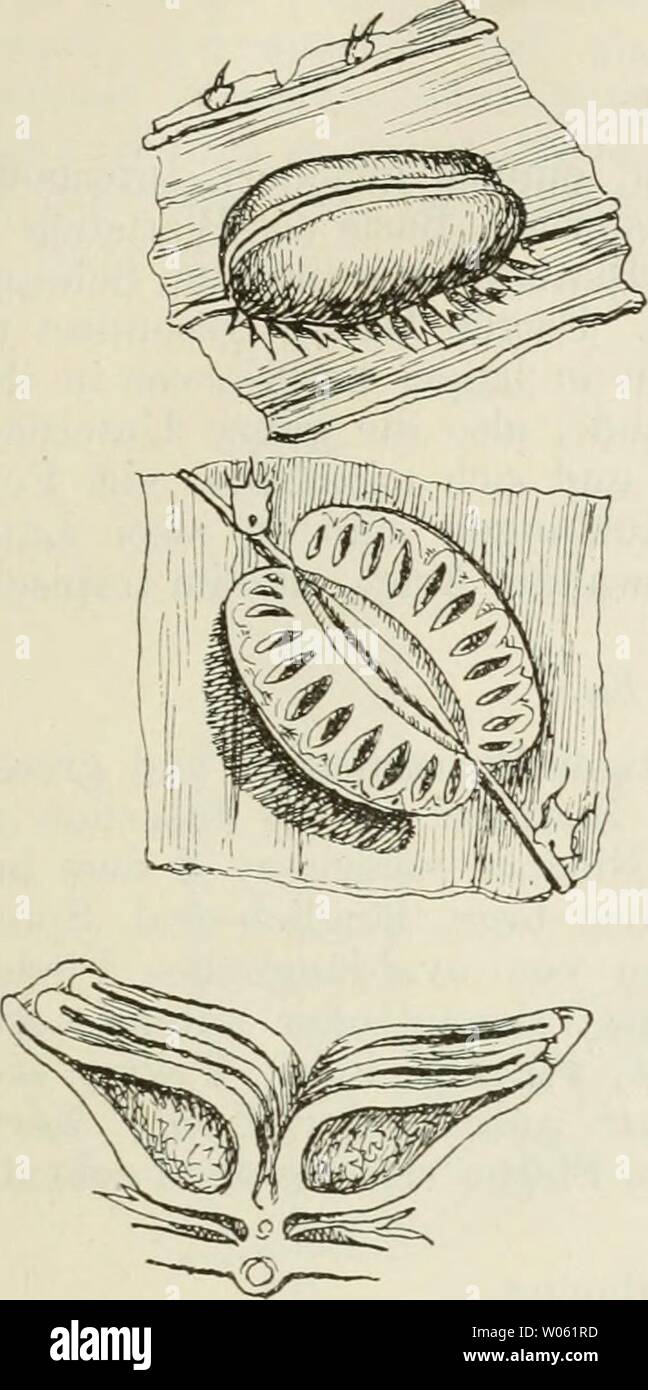
RMW061RD–Archive image from page 374 of Die Farnkräuter der Erde . Die Farnkräuter der Erde : beschreibende Darstellung der geschlechter und wichtigeren Arten der Farnpflanzen mit besonderer Berücksichtigung der Exotischen diefarnkruterd00chriuoft Year: 1897 Marattia Sm. 359 Hab. Diese an das Blatt von Weinmannia erinnernde Art ist in den Antillen und Central- Amerika zu Hause, und es liegt eine nicht verschieden erscheinende Pflanze von Celebes (1. Sarasin) vor. Nahe steht ihr auch eine Form der Sandwichs-Inseln (M. Douglasii Bak.).
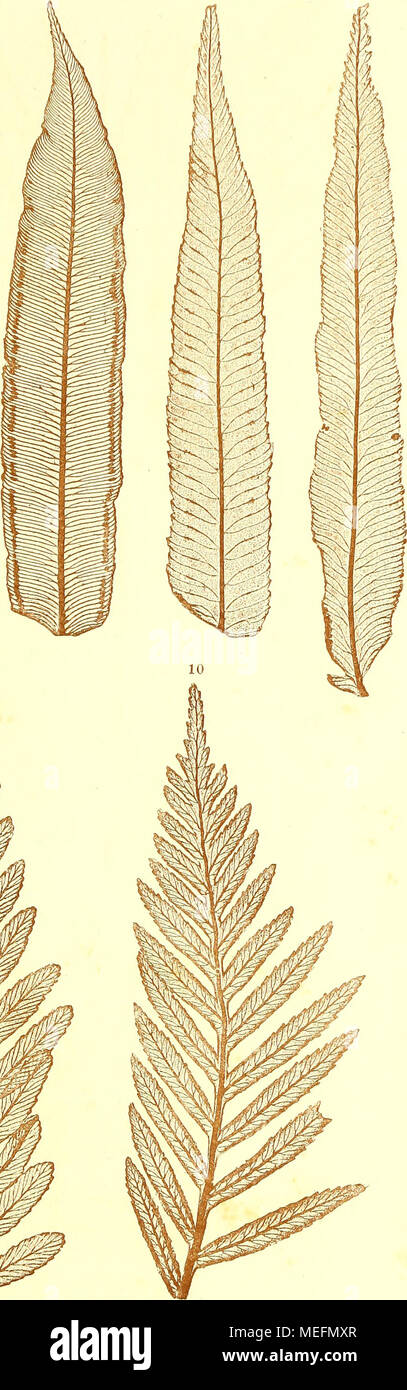
RMMEFMXR–. Die farnkräuter der jetztwelt, zur untersuchung und bestimmung, der in den formationen der erdrinde eingeschlossenen überreste von vorweltlichen arten dieser ordnung nach dem flächen-skelet . 1, 2. Marattia Kaulfnssii J. Smith. 3. Angiojiteris sp. (Mayotte). 4. Marattia Baddiana Schott. 5. 6. Psilodochea salicifolia P r e s 1.
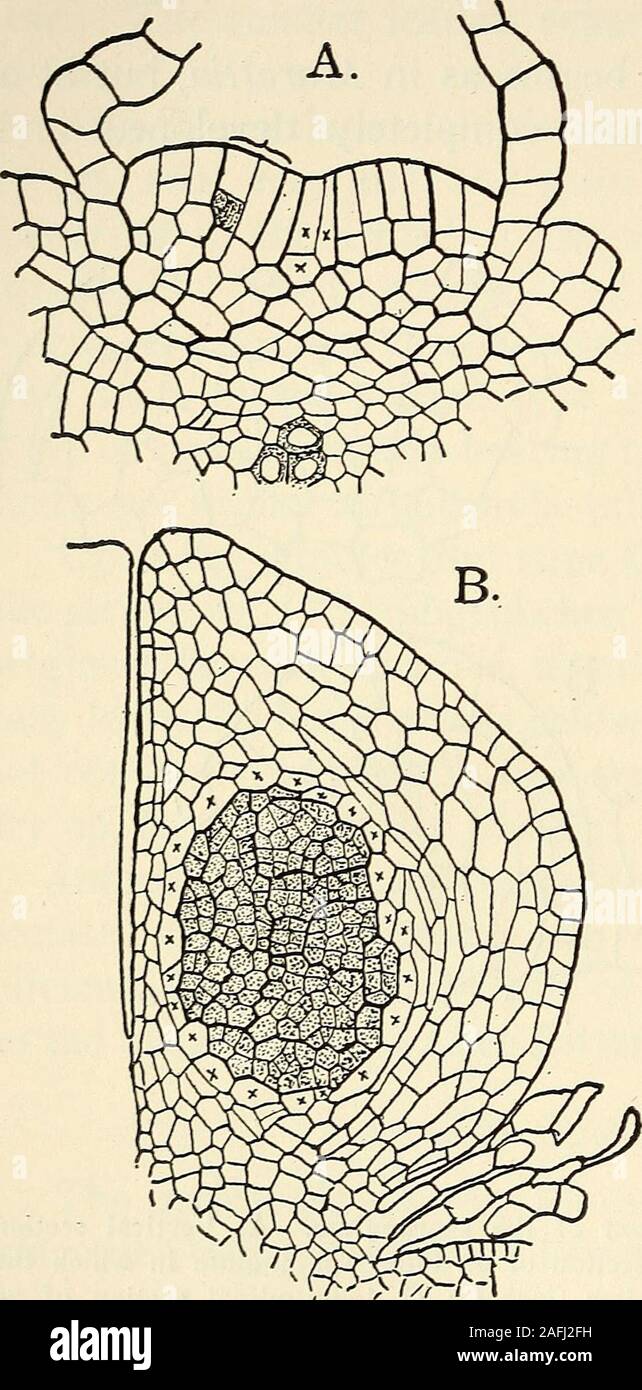
RM2AFJ2FH–. The structure and development of mosses and ferns (Archegoniatae). The top of thesporangium and the innerwall are composed of but onelayer of cells (exclusive ofthe tapetum), which are flatand more delicate than thoseupon the outer side. Nearthe top on its outer side is atransverse line of cells withthickened darker walls, whichproject somewhat above thelevel of the others. This isT7 , ,. ^. ^ . , ^ the annulus or ring, and re- xiG. 165.—Marattia fraxmea. A, Transverse ° section of young synangium, X225; B, SCmblcS cloSCly that of Os- similar section of an older synangium, ^^i^j^da. Lining t

RMPG00NT–. Lessons in botany. Botany. Fig. 148. Fertilization in a fern (marattia). sp, spermato-. Fig. 149. Voting plant of Pteris serrulata still attached to prothallium. when they are Campbell.) swimming around over the under surface of the prothallium. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Atkinson, George Francis, 1854-1918. New York, H. Holt and company
![Archive image from page 821 of Dictionnaire d'horticulture illustré par. Dictionnaire d'horticulture illustrà / par D. Bois prÃface de Maxime Cornu avec la collaboration de E. Andrà ... [et al.]. . dictionnairedhor00bois Year: 1893 MARATTIA. 810 MARCOTTAGE. sores marginaux, entourÃs inft. d'un involucre fimbriÃ. Serre chaude. M. Kaulfussii J. Sm. [Fig. 561.) â Antilles, AmÃr. mÃr. â Frondes quadripinnatifides, d'en- viron 1 m. de hauteur. E. R. MARCESCENT. Se dit des organes qui se flÃ- trissent et se dessèchent après avoir atteint leur dÃveloppement, mais qui sont persistants pendant Stock Photo Archive image from page 821 of Dictionnaire d'horticulture illustré par. Dictionnaire d'horticulture illustrà / par D. Bois prÃface de Maxime Cornu avec la collaboration de E. Andrà ... [et al.]. . dictionnairedhor00bois Year: 1893 MARATTIA. 810 MARCOTTAGE. sores marginaux, entourÃs inft. d'un involucre fimbriÃ. Serre chaude. M. Kaulfussii J. Sm. [Fig. 561.) â Antilles, AmÃr. mÃr. â Frondes quadripinnatifides, d'en- viron 1 m. de hauteur. E. R. MARCESCENT. Se dit des organes qui se flÃ- trissent et se dessèchent après avoir atteint leur dÃveloppement, mais qui sont persistants pendant Stock Photo](https://c8.alamy.com/comp/W1D0EG/archive-image-from-page-821-of-dictionnaire-dhorticulture-illustr-par-dictionnaire-dhorticulture-illustr-par-d-bois-prface-de-maxime-cornu-avec-la-collaboration-de-e-andr-et-al-dictionnairedhor00bois-year-1893-marattia-810-marcottage-sores-marginaux-entours-inft-dun-involucre-fimbri-serre-chaude-m-kaulfussii-j-sm-fig-561-antilles-amr-mr-frondes-quadripinnatifides-den-viron-1-m-de-hauteur-e-r-marcescent-se-dit-des-organes-qui-se-fl-trissent-et-se-desschent-aprs-avoir-atteint-leur-dveloppement-mais-qui-sont-persistants-pendant-W1D0EG.jpg)
RMW1D0EG–Archive image from page 821 of Dictionnaire d'horticulture illustré par. Dictionnaire d'horticulture illustrà / par D. Bois prÃface de Maxime Cornu avec la collaboration de E. Andrà ... [et al.]. . dictionnairedhor00bois Year: 1893 MARATTIA. 810 MARCOTTAGE. sores marginaux, entourÃs inft. d'un involucre fimbriÃ. Serre chaude. M. Kaulfussii J. Sm. [Fig. 561.) â Antilles, AmÃr. mÃr. â Frondes quadripinnatifides, d'en- viron 1 m. de hauteur. E. R. MARCESCENT. Se dit des organes qui se flÃ- trissent et se dessèchent après avoir atteint leur dÃveloppement, mais qui sont persistants pendant
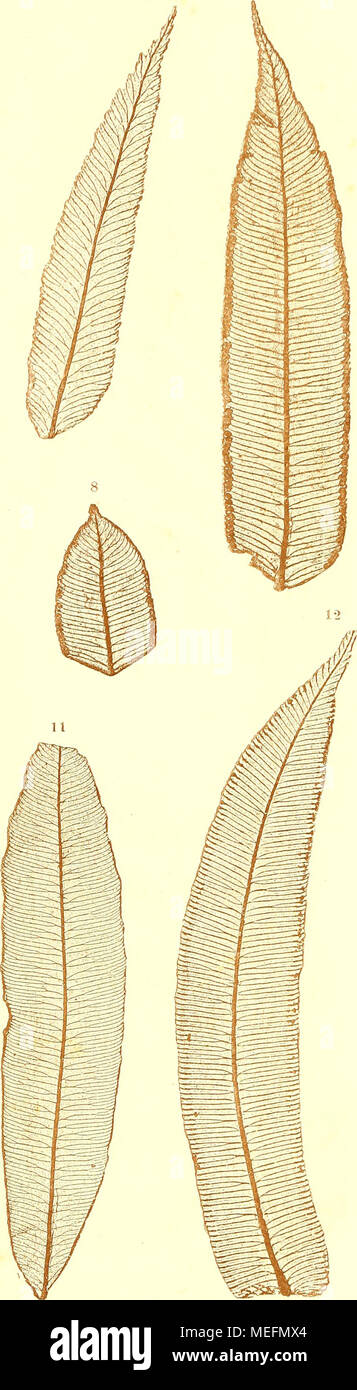
RMMEFMX4–. Die farnkräuter der jetztwelt, zur untersuchung und bestimmung, der in den formationen der erdrinde eingeschlossenen überreste von vorweltlichen arten dieser ordnung nach dem flächen-skelet . 7, 8. Marattia microcarpa Mett. 9. 10. Todea africana WiUd. 11. Angioineris latifolia Presl. 12. Amjiopteris evecta Hoffm. Kms der k. k. l[of- iin.l Sla.its.lnicitorni
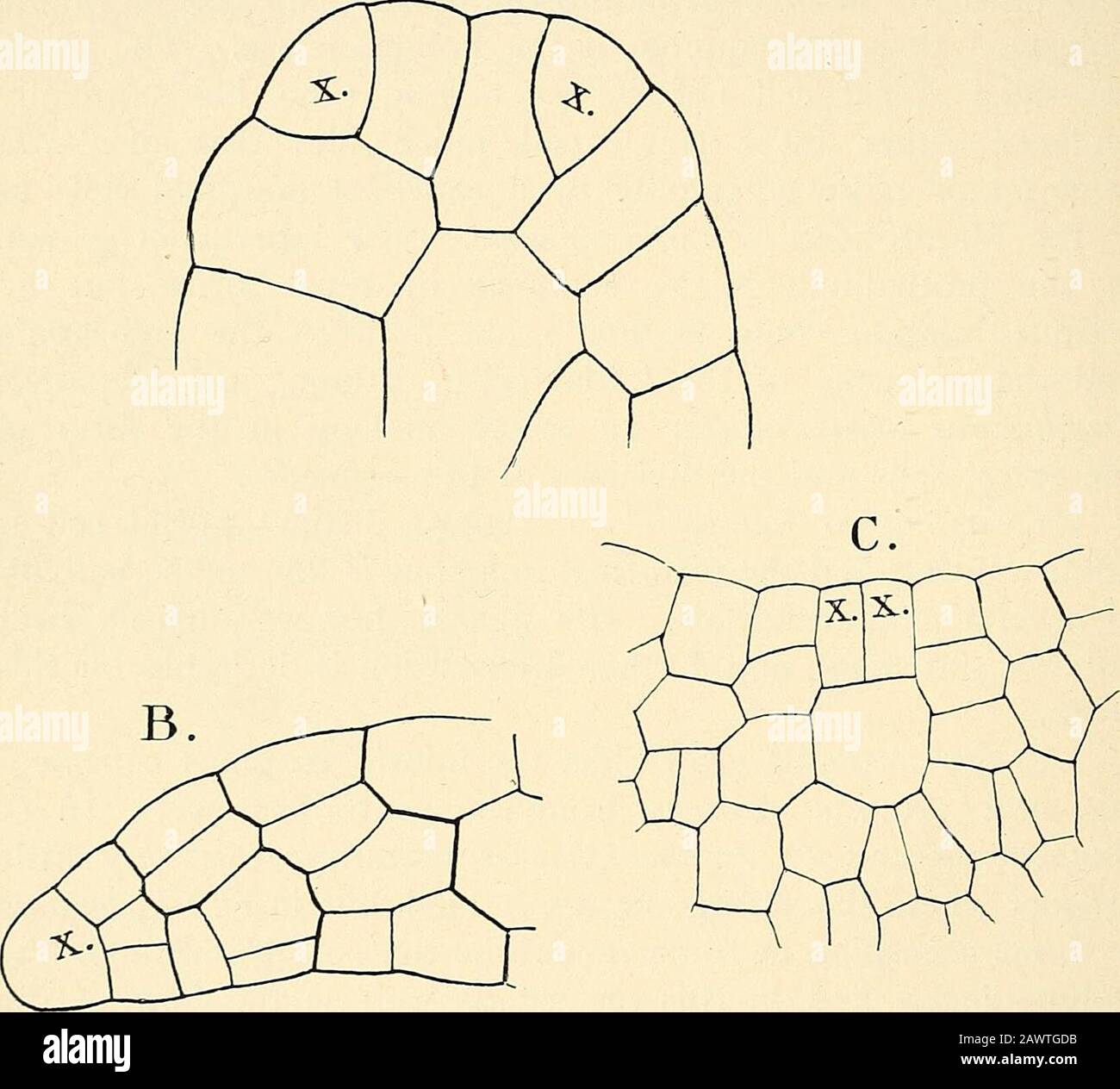
RM2AWTGDB–The structure & development of the mosses and ferns (Archegoniatae) . and in this respect, as well as the form of theapical cells, seems to approach the latter. In this species theprothallium is lighter coloured, and the root-hairs not so dark,while in its dark green colour and fleshy texture 0. cinnainomearecalls AntJioceros IcEvis or Marattia. Where a cell mass is formed at first, this condition istemporary, and an apical cell is established which gives rise tothe ordinary flat prothallium. The small male prothallia, whichare formed in large numbers, exhibit various irregularities, andquite
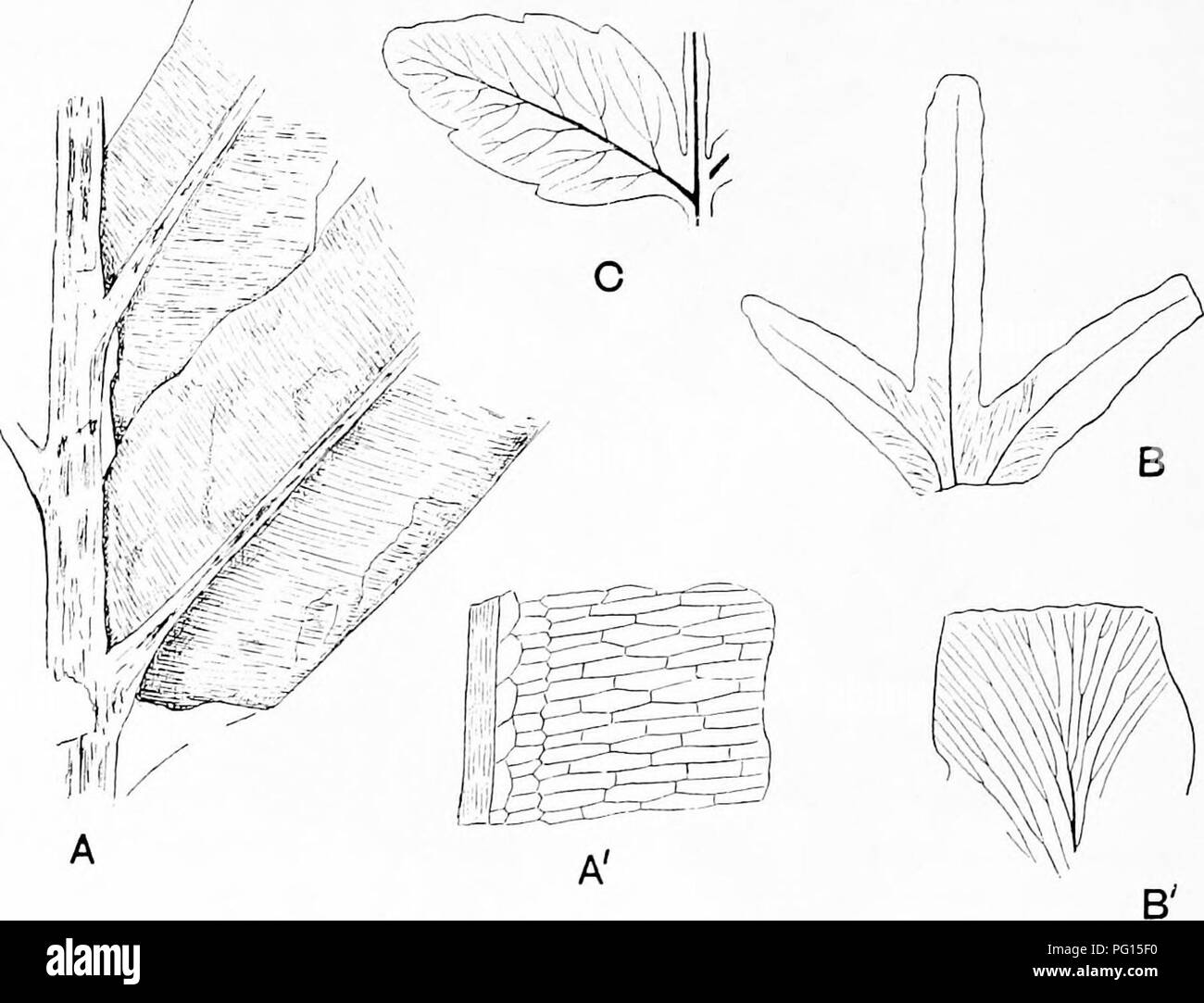
RMPG15F0–. Fossil plants : for students of botany and geology . Paleobotany. 350 FILICALES [CH. Ruffordia Goepperti (Dunk.). Fig. 260. This Wealden fern^ has been doubtfully assigned to the Schizaeaceae on the ground of the resemblance of the sterile fronds to those of some species of Aneimia, and because of the difference between the sterile and fertile pinnae (Fig. 260). Ruffordia cannot be regarded as a well authenticated member of the Schizaeaceae.. Fig. 261. A, A'. Chrysodium lanzaeanum. B, B'. Lygudium Kaulfussi. C. Marattia Hookeri. (After Gardner and Ettingshauaen ; A, B, | nat. size.) Lygodium
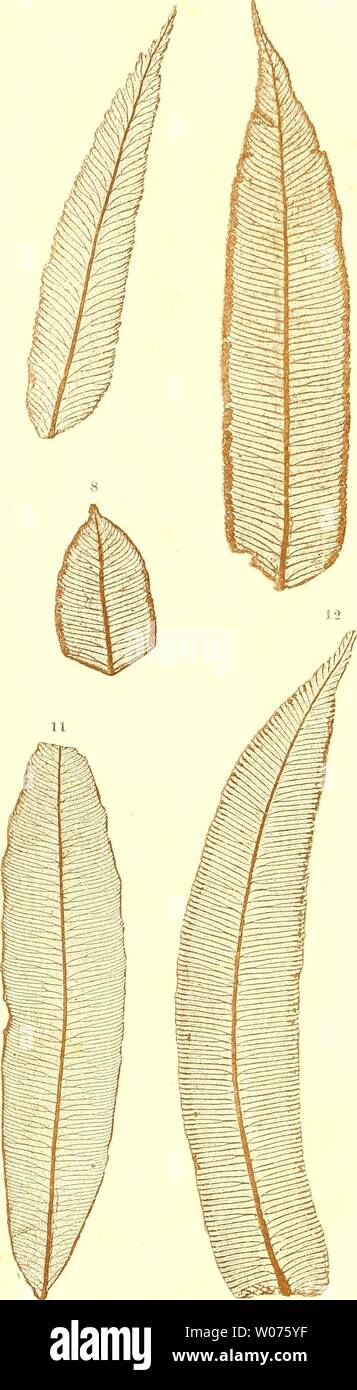
RMW075YF–Archive image from page 500 of Die farnkräuter der jetztwelt, zur. Die farnkräuter der jetztwelt, zur untersuchung und bestimmung, der in den formationen der erdrinde eingeschlossenen überreste von vorweltlichen arten dieser ordnung nach dem flächen-skelet diefarnkruterder00etti Year: 1865 1, 2. Marattia Kaulfnssii J. Smith. 3. Angiojiteris sp. (Mayotte). 4. Marattia Baddiana Schott. 5. 6. Psilodochea salicifolia P r e s 1. 7, 8. Marattia microcarpa Mett. 9. 10. Todea africana WiUd. 11. Angioineris latifolia Presl. 12. Amjiopteris evecta Hoffm. Kms der k. k. l[of- iin.l Sla.its.lnicitorni
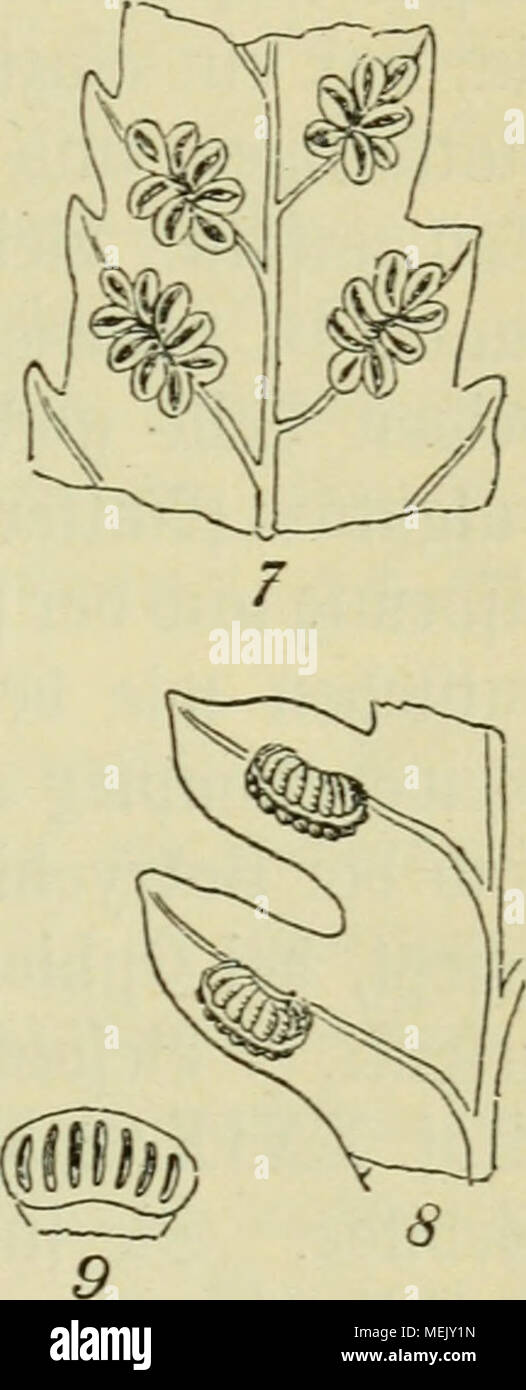
RMMEJY1N–. Die Algen, Moose und Farnpflanzen. . £af. XXIX. 1. Botrychium lunaria. 2. 2>e»gt., ^rotfjattiumquericfnutr, mit Slnttjertbien, ?trd)egonium unb (Jmbrrjo. 3. 2)e3gl., ^rotfjaüium; siuei (Jmbrt)* ouen trei6en if)re SBursein Ijeruor. 4. Ophioglossum vulgatum. 5. 2)e3gl. Gpi&e be3 fevtilen 23tattei(§. G. Aspidium cristatum, Verlauf ber Ö5efö»> bünbet in ber 8(cbi'e, nur bie 3>orberfeite bargeftellt. 7. Angiopteris caudata, SötattftüdE mit <Sori§. 8. Marattia fpec, Stattftüd mit <Sori§. 9. ©eägt«» burdn'crjnittener @oru§. afrifa bi§ Sßofinteften) finb fie gu einem merjrfäcrjeri
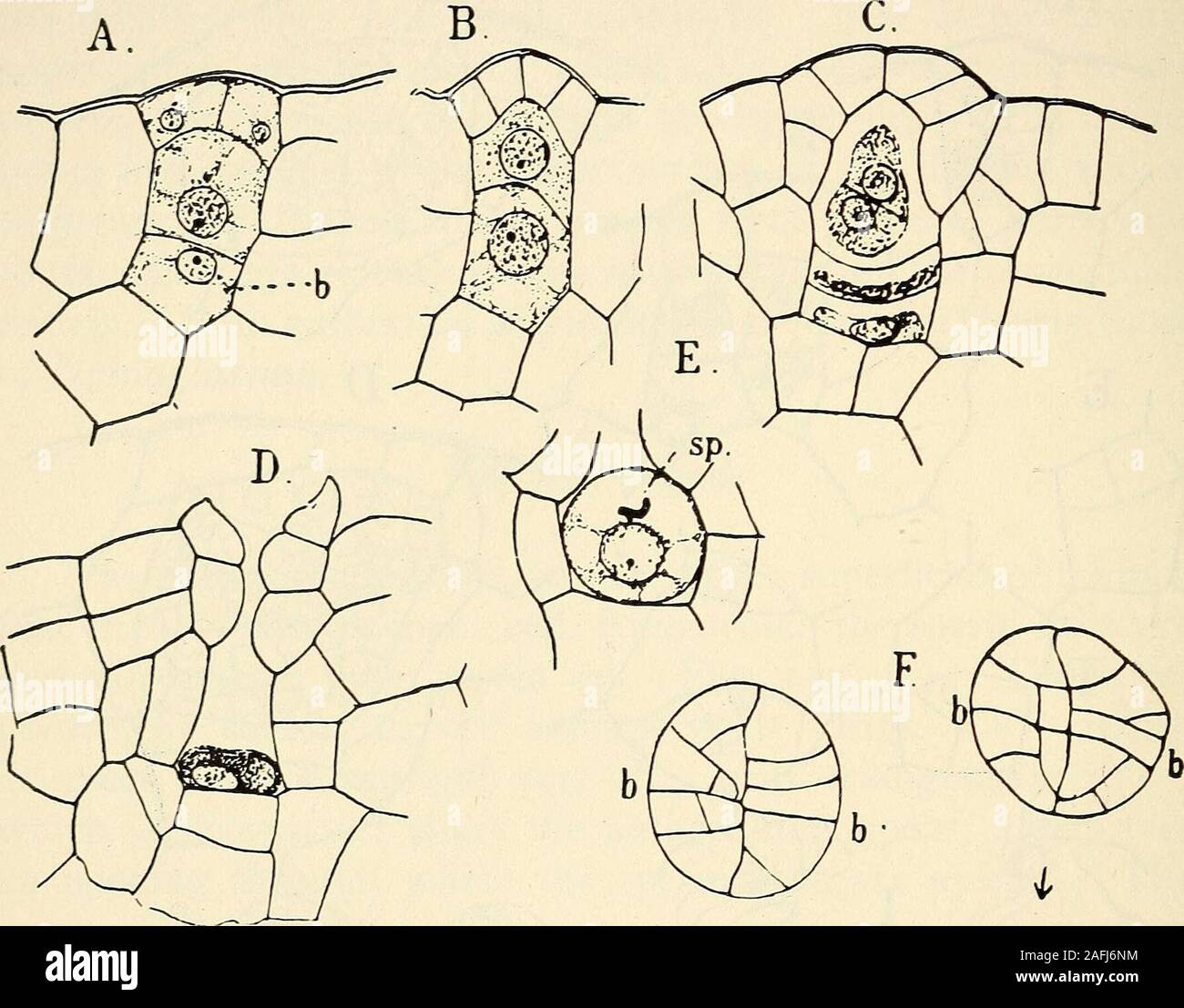
RM2AFJ6NM–. The structure and development of mosses and ferns (Archegoniatae). point, and apparently any superficial cell ispotentially an archegonium mother cell. The latter dividesusually into three superimposed cells (Fig. 153, A), of whichthe lowest (b) forms the base of the archegonium. The basalcell, however, may be absent in Marattia Douglasii, as is alsothe case in Angiopteris and Dancea. From the middle cell by atransverse division are formed the primary neck canal cell and 280 MOSSES AND FERNS CHAP. the central cell. Each of these divides again transversely. Inthe upper one this division is of
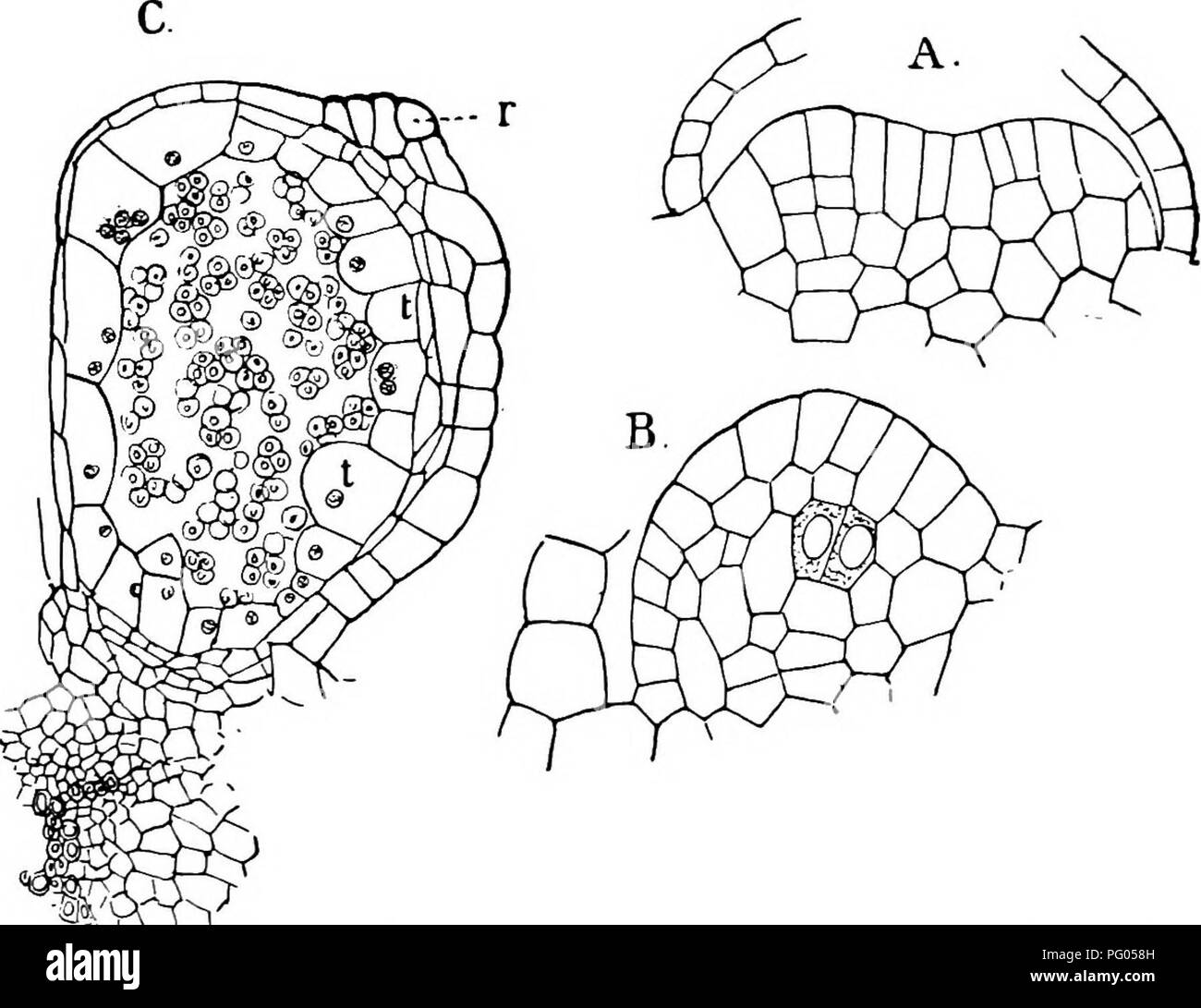
RMPG058H–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. MARATTIALES 293 between. In each half there are differentiated the separate archesporial groups of cells corresponding to the separate chambers found in the complete synangium." The whole process takes, according to his account, about six months. Luerssen was unable either in Marattia or Angiopteris to trace back the archesporium to a single cell, which Goebel (3) claims is present in the latter. In Angiopteris the process begins as in Marattia^ but at a period when the leaf is almost com

RMMCRB5G–. A) Angioptciis Willinkii: 1) :gabituS, ftart üertloincrt; 2) fevtitc a^tattfieber, non unten; 3) Sorus, ftaiE i>ei-= grö^ert. 3(06.47: 2)larattia = gante (Marattiaeeae). B) Danaea i'lliptica: 1) JertUe 1 gottite aj^attfieber, von un^ SJIattficbcr, uon unten; 2) jiuei Sori, ocrgröpert; 3) ein iSoruä im Duerfdinitt. C) Marattia fraxinifolia: 1) ten; 2) Seil einer aiattfiebev mit ju'ei Sori, »ergröpert; 3)Sornä im C.ucrfd)nitt, iicr= gröjjcrt. D) Marattia weinuiamiiifolia: 1) Seit bes aSIatteä; 2) §ieber. E) Kaulfassia aesculifolia: 1) Jötottftüct; 2) Soru^, oergr.; 3) Soruä im xluerfc^nitt,
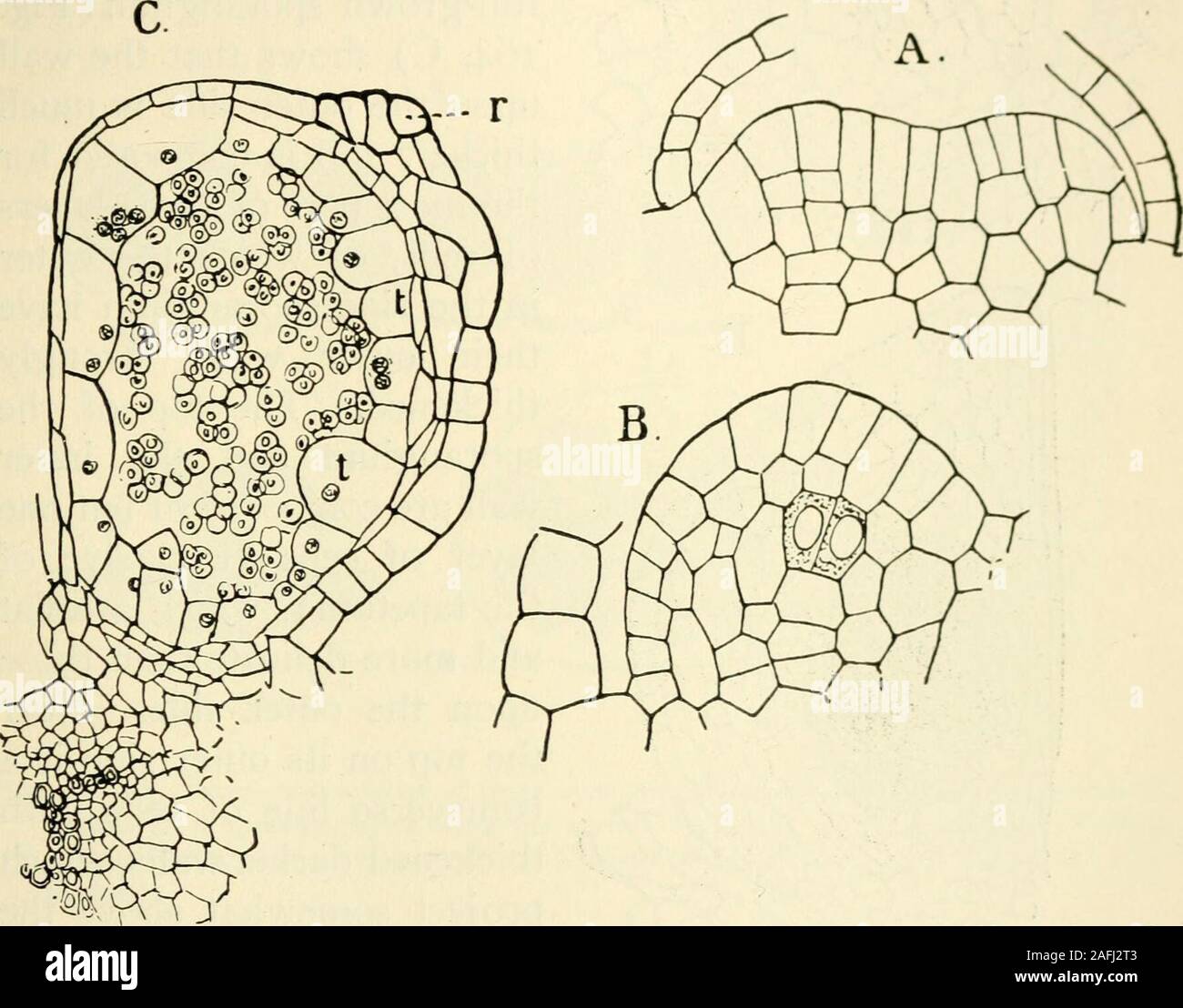
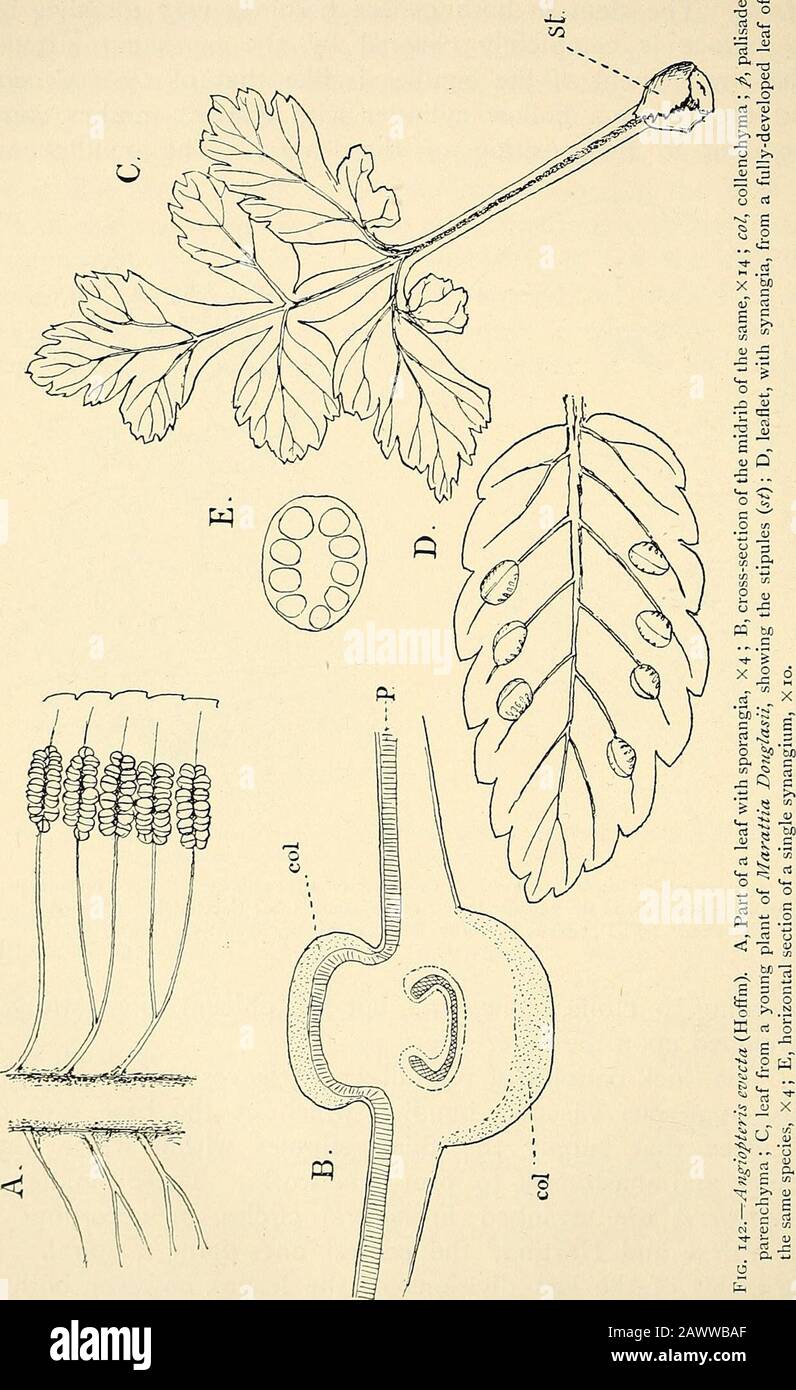
RM2AFJ2T3–. The structure and development of mosses and ferns (Archegoniatae). s : Tn Marattia the first differentiation of the spo-rangium begins while the young leaf is still rolled up between thestipules of the next older one. The tissue above the fertile veinis more strongly developed than the adjoining parenchyma, andforms an elevated cushion parallel with the vein. This is thereceptacle, which develops two parallel ridges, separated by acleft. These two ridges grow up until they meet, and theiredges grow together and completely close the cleft which lies VIII MARATTIALES 293 between. In each half
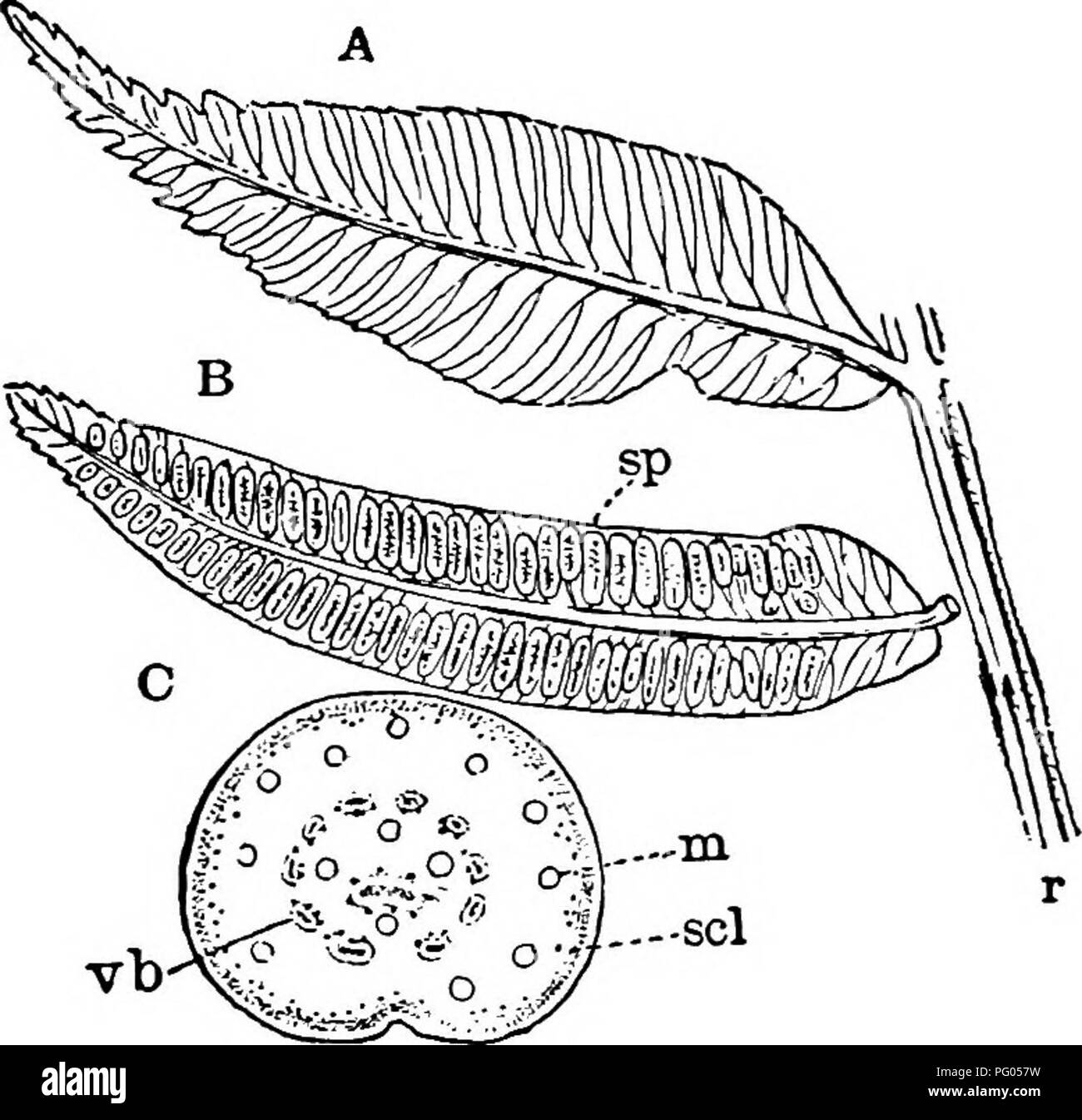
RMPG057W–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. MARATTIALES 299 Marattia includes about twelve species of tropical and sub- tropical Ferns, both of the Old World and the New. Kaul- fiissia includes but a single species, belonging to southeastern Asia. The synangia are scattered over the lower surface of the palmate leaf, and are circular, with a central space into which the separate loculi open by a slit, as in Marattia. Kaul- fussia is characterised by very large pores upon the lower side of the leaf. A study of the development of these sh
![. Dictionnaire d'horticulture illustré / par D. Bois préface de Maxime Cornu avec la collaboration de E. André ... [et al.]. . Fig. 56L. â Marattia Kaulfussii J. Sm. MARATTIA Smith. (Fougères, Marattiacées.) Plantes à frondes bi- ou tripinnées, portant inft. des sores constitués par des sporanges soudés, sur deux rangs, en un sporange unique, bivalve ou cyathiforme, à déhiscence fissurale. M. alata Sm. â Hook. Gen. Fil. 26. â Amér. trop. â Rhizome court. Frondes tripinnées, d'environ 50 cm., à stipe peu écailleux, à pin- nules oblongues, cunéiformes à la base, denti- culées, po Stock Photo . Dictionnaire d'horticulture illustré / par D. Bois préface de Maxime Cornu avec la collaboration de E. André ... [et al.]. . Fig. 56L. â Marattia Kaulfussii J. Sm. MARATTIA Smith. (Fougères, Marattiacées.) Plantes à frondes bi- ou tripinnées, portant inft. des sores constitués par des sporanges soudés, sur deux rangs, en un sporange unique, bivalve ou cyathiforme, à déhiscence fissurale. M. alata Sm. â Hook. Gen. Fil. 26. â Amér. trop. â Rhizome court. Frondes tripinnées, d'environ 50 cm., à stipe peu écailleux, à pin- nules oblongues, cunéiformes à la base, denti- culées, po Stock Photo](https://c8.alamy.com/comp/MEK1JG/dictionnaire-dhorticulture-illustr-par-d-bois-prface-de-maxime-cornu-avec-la-collaboration-de-e-andr-et-al-fig-56l-marattia-kaulfussii-j-sm-marattia-smith-fougres-marattiaces-plantes-frondes-bi-ou-tripinnes-portant-inft-des-sores-constitus-par-des-sporanges-souds-sur-deux-rangs-en-un-sporange-unique-bivalve-ou-cyathiforme-dhiscence-fissurale-m-alata-sm-hook-gen-fil-26-amr-trop-rhizome-court-frondes-tripinnes-denviron-50-cm-stipe-peu-cailleux-pin-nules-oblongues-cuniformes-la-base-denti-cules-po-MEK1JG.jpg)
RMMEK1JG–. Dictionnaire d'horticulture illustré / par D. Bois préface de Maxime Cornu avec la collaboration de E. André ... [et al.]. . Fig. 56L. â Marattia Kaulfussii J. Sm. MARATTIA Smith. (Fougères, Marattiacées.) Plantes à frondes bi- ou tripinnées, portant inft. des sores constitués par des sporanges soudés, sur deux rangs, en un sporange unique, bivalve ou cyathiforme, à déhiscence fissurale. M. alata Sm. â Hook. Gen. Fil. 26. â Amér. trop. â Rhizome court. Frondes tripinnées, d'environ 50 cm., à stipe peu écailleux, à pin- nules oblongues, cunéiformes à la base, denti- culées, po
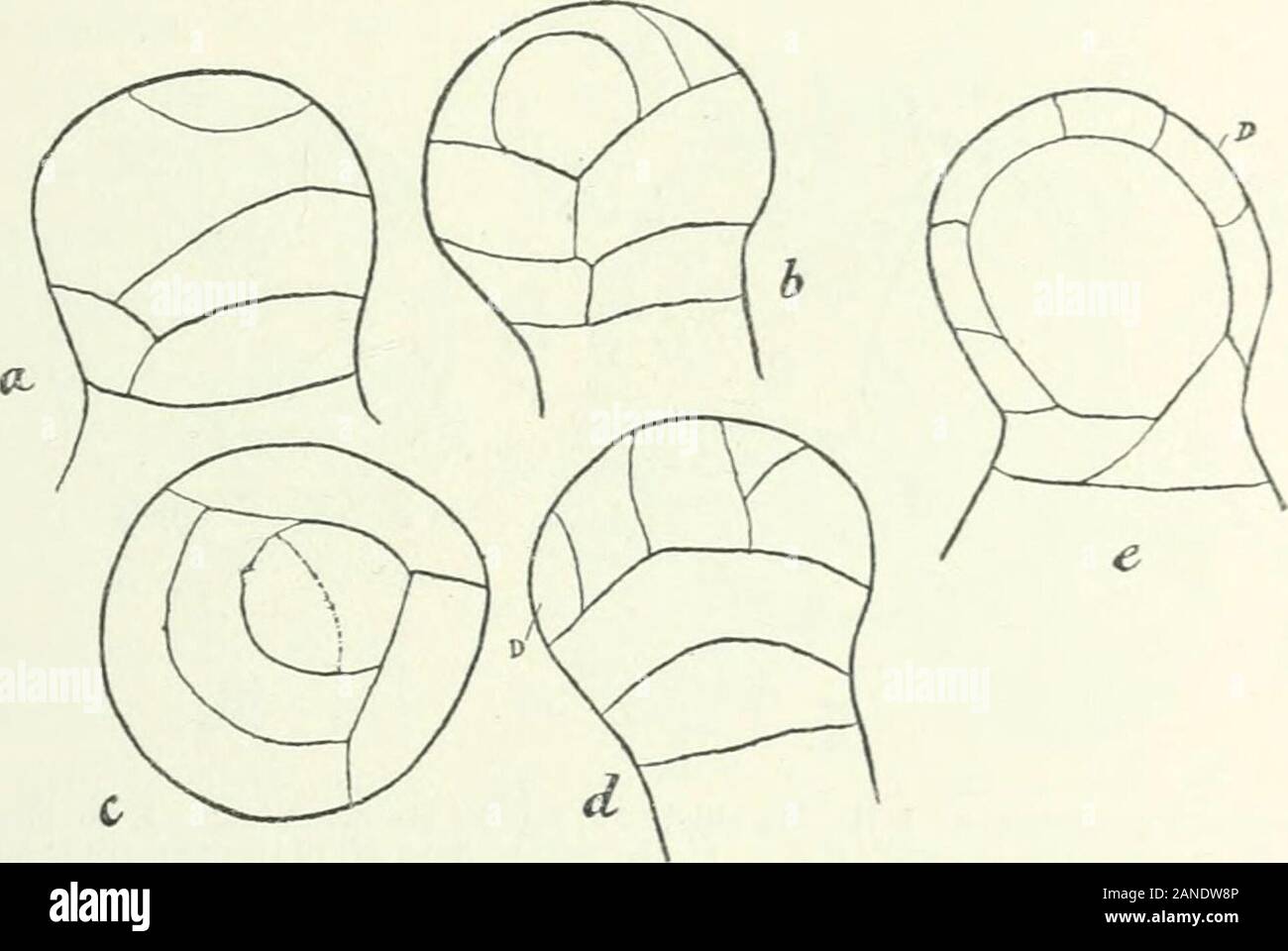
RM2ANDW8P–Organography of plants, especially of the archegoniatae and spermaphyta . htca Eotanica, viii (1SS7), discusses my views. DEVELOPMENT OF THE ANTHERIDIUM 179 greatly of what we find in the primordium of the antheridium of theOsmundaceae (Figs. 133, III ; 134)- Osmunda. In Osmunda (Fig. 134) many cell-walls arise, inclined inthree directions in space, which lead to the formation of an antheridial stalk ;then follows a wall curved in a cap-like manner which corresponds withthat which in Marattia, Equisetum, and Lycopodium, separates the opercularlayer from the cell within, and then, by further di
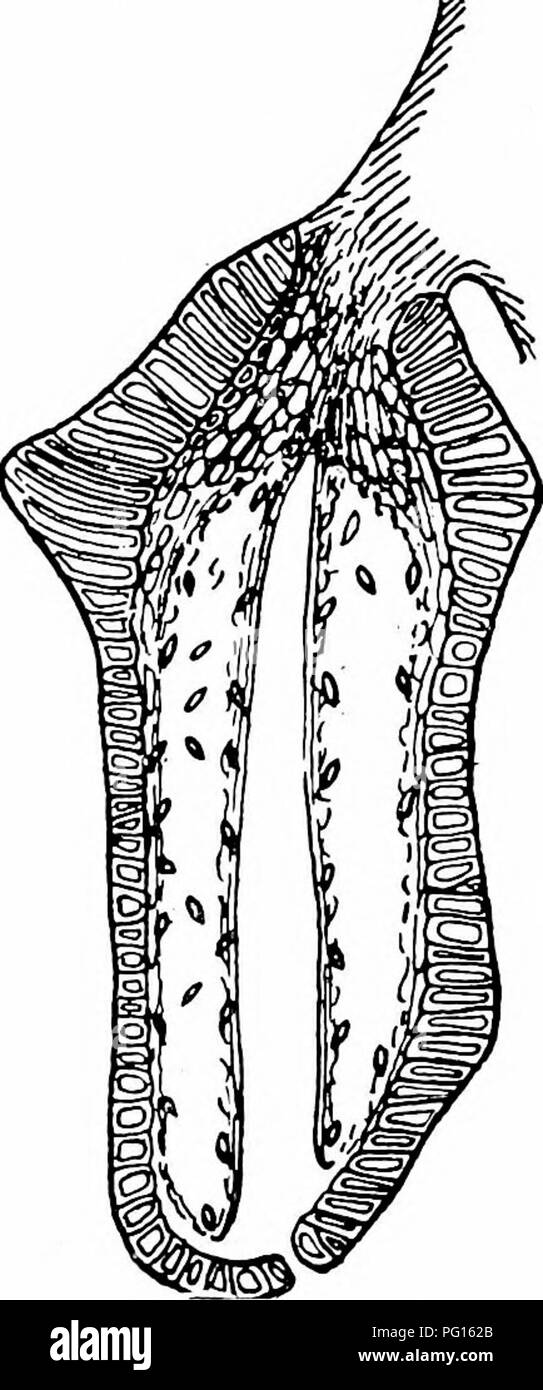
RMPG162B–. Studies in fossil botany . Paleobotany. BENNETTITEAE 537 in the case of unreduced sporophylls, is often departed from. The shortly-stalked synangia are much broader than long, contain two rows of loculi (Figs. 210 and 211), and closely resemble those of certain Marattiaceae, simulating in a remarkable degree the stalked synangia of Marattia Kaulfussii} The analog)^ of this species, though not of any special taxonomic significance, shows that there is no absolute necessity to regard the pedicellate syn- angia of the Bennettiteae as representing distinct leaflets. The form of the synangia in C
RM2AWWBAF–The structure & development of the mosses and ferns (Archegoniatae) . ^very numerous vascular bundles, which at the base give offbranches that supply the thick stipules within which theybranch and anastomose to form a network. These bundles inA^igiopteris are arranged in several circles, or according toDe Vriese and Harting,^ the central ones form a spiral. Inthe rachis of the last divisions of the leaves, however, both of ^ Bower (2), p. 579. - Holle (2), p. 217. ^ De Vriese (i). 270 MOSSES AND FERNS CHAP. Marattia and Angiopteris, there is but a single axial bundle, asin the petiole of the c
![. Fossil plants : for students of botany and geology . Paleobotany. XXVll] APHLEBIA 527 or stipular structures which cannot "be connected with definite species of fronds. It is clear that some Aphlebiod leaflets, such as those of Dactylotheca, served as protective organs for the. Fig. 351. Scale-leaf of Gunnera manicata. (Slightly reduced. M.S.) unexpanded pinnae', and in all probability the large Aphebiae served the same purpose as the fleshy stipules of Angiopteris and Marattia which cover the uncoiled fronds. The pinnatifid 1 See p. 406, fig. 293; Potonie (03) also figures a young fron Stock Photo . Fossil plants : for students of botany and geology . Paleobotany. XXVll] APHLEBIA 527 or stipular structures which cannot "be connected with definite species of fronds. It is clear that some Aphlebiod leaflets, such as those of Dactylotheca, served as protective organs for the. Fig. 351. Scale-leaf of Gunnera manicata. (Slightly reduced. M.S.) unexpanded pinnae', and in all probability the large Aphebiae served the same purpose as the fleshy stipules of Angiopteris and Marattia which cover the uncoiled fronds. The pinnatifid 1 See p. 406, fig. 293; Potonie (03) also figures a young fron Stock Photo](https://c8.alamy.com/comp/PG14GG/fossil-plants-for-students-of-botany-and-geology-paleobotany-xxvll-aphlebia-527-or-stipular-structures-which-cannot-quotbe-connected-with-definite-species-of-fronds-it-is-clear-that-some-aphlebiod-leaflets-such-as-those-of-dactylotheca-served-as-protective-organs-for-the-fig-351-scale-leaf-of-gunnera-manicata-slightly-reduced-ms-unexpanded-pinnae-and-in-all-probability-the-large-aphebiae-served-the-same-purpose-as-the-fleshy-stipules-of-angiopteris-and-marattia-which-cover-the-uncoiled-fronds-the-pinnatifid-1-see-p-406-fig-293-potonie-03-also-figures-a-young-fron-PG14GG.jpg)
RMPG14GG–. Fossil plants : for students of botany and geology . Paleobotany. XXVll] APHLEBIA 527 or stipular structures which cannot "be connected with definite species of fronds. It is clear that some Aphlebiod leaflets, such as those of Dactylotheca, served as protective organs for the. Fig. 351. Scale-leaf of Gunnera manicata. (Slightly reduced. M.S.) unexpanded pinnae', and in all probability the large Aphebiae served the same purpose as the fleshy stipules of Angiopteris and Marattia which cover the uncoiled fronds. The pinnatifid 1 See p. 406, fig. 293; Potonie (03) also figures a young fron
RM2AFHJ3T–. The structure and development of mosses and ferns (Archegoniatae). n colour and fleshy texture 0. cin-namomea recalls Anfhoceros Iccvis or Marattia. Where a cell mass is formed at first, this condition is tem-porary, and an apical cell is established which gives rise to theordinary flat prothallium. The small male prothallia, which areproduced in large numbers, exhibit various irregularities andquite commonly do not show any definite apical growth, and inO. Claytoniana especially often branch irregularly, or in somecases there is a true dichotomy (Fig. 193, A.) Slender fila-mentous prothalli
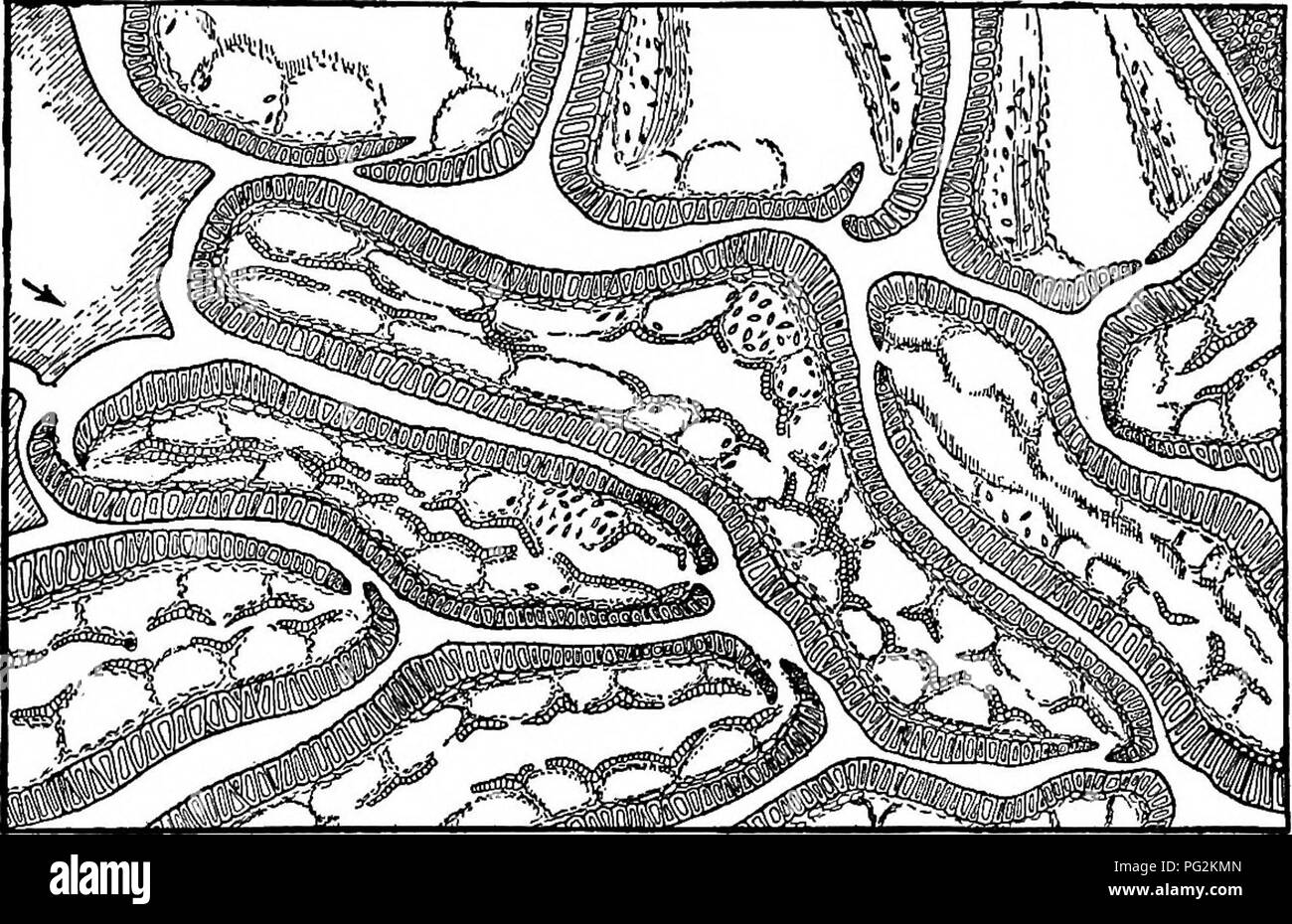
RMPG2KMN–. Morphology of gymnosperms. Gymnosperms; Plant morphology. BENNETTITALES 77 which project only i. 5 cm. beyond the disk, each bearing two lat- eral rows of synangia along the rachis. This remarkable "reduction" of the stamen set, as compared with that in Cycadeoidea, has inten- sified the suggestion of an angiosperm connection. That the stamens of Bennettitales bore synangia of the ordinary Marattia type (fig. 61) has been shown to be true of all the forms. Fig. 61.—Cycadeoidea dacotensis: part of transverse section through unexpanded microsporophyll, showing synangia with loculi ar
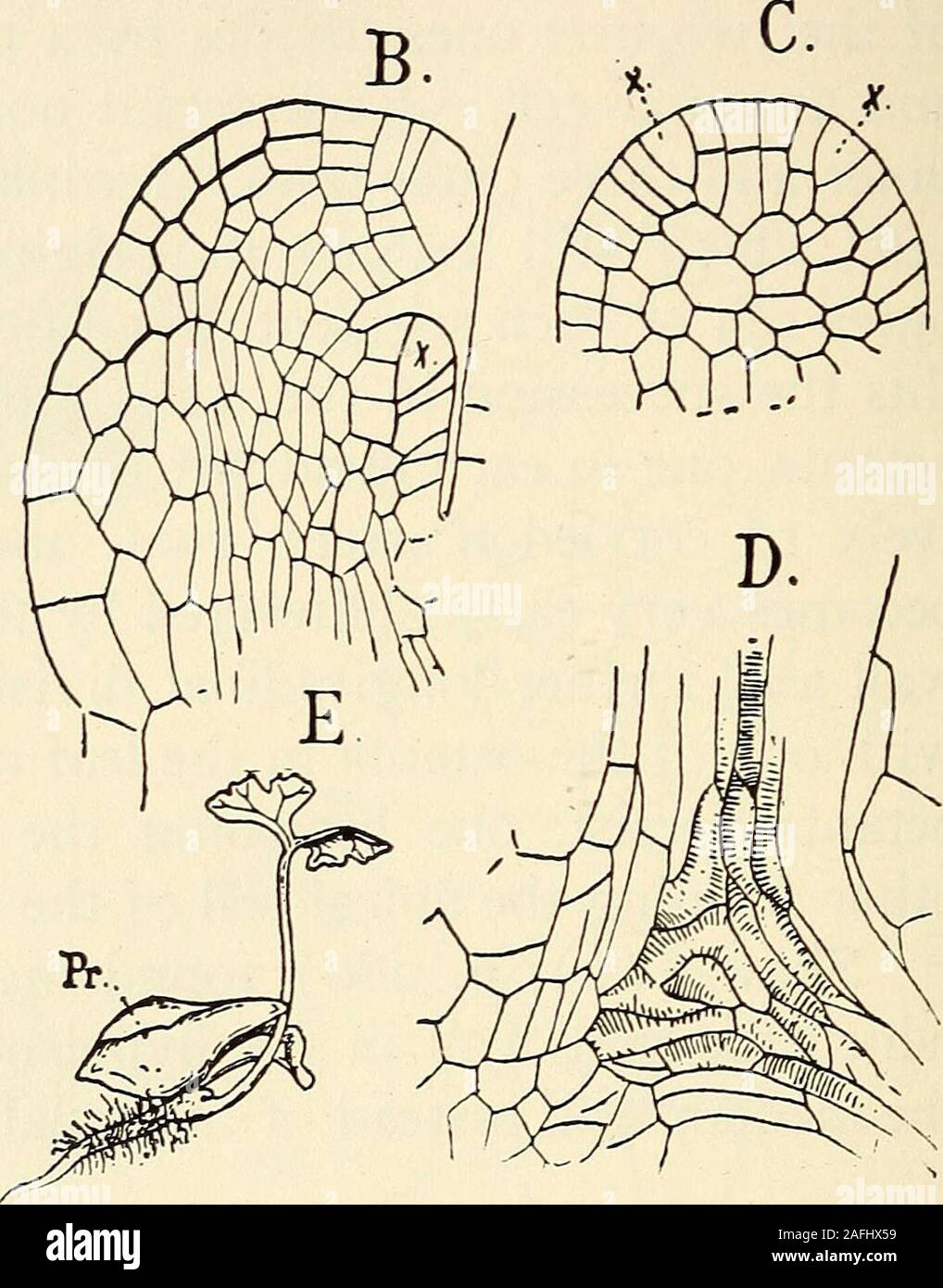
RM2AFHX59–. The structure and development of mosses and ferns (Archegoniatae). Fig. 179.—Onoclea struthiopteris. A, Longitudinal section of young sporophyte stillconnected with the prothallium (Pr), X6o; B, the apex of same, Xi8o; C, surfaceview of the young cotyledon showing the first dichotomy; D, central region of A,showing the primary tracheary tissue, Xi8o; E, young sporophyte with nearlyfull-grown cotyledon and primary root, X3; st, stem; L^, cotyledon; L^ secondleaf; F, foot; Pr, prothallium. the prothallium. As in Marattia the growth is much strongerupon the outer side and the leaf is strongly c
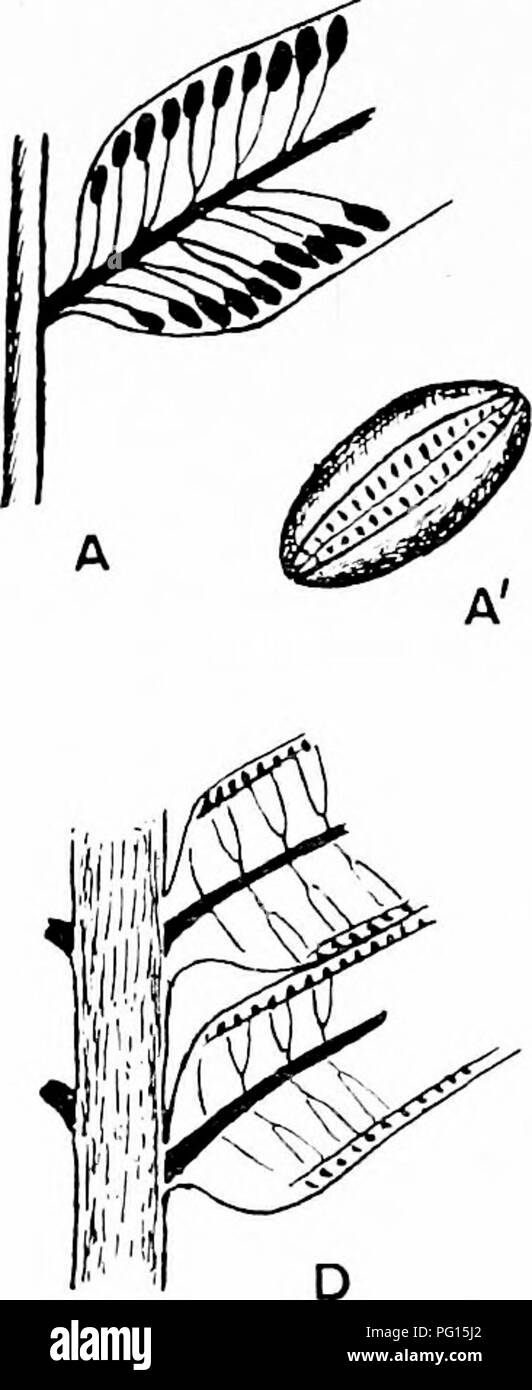
RMPG15J2–. Fossil plants : for students of botany and geology . Paleobotany. 320 FILICALES [CH. but the sori agree with those of Angiopteris except in their greater length and in the larger number of sporangia. Marattia. This genus, which extends " all round the world within the tropics 1," includes some species which closely resemble Angiopteris, while others are characterised by more finely divided leaves with smaller ultimate segments. The fleshy stipules occasionally have an irregularly pinnatifid form (fig. 241, B). The sporangia are represented by oval synangia^ (fig. 245, A ; the black
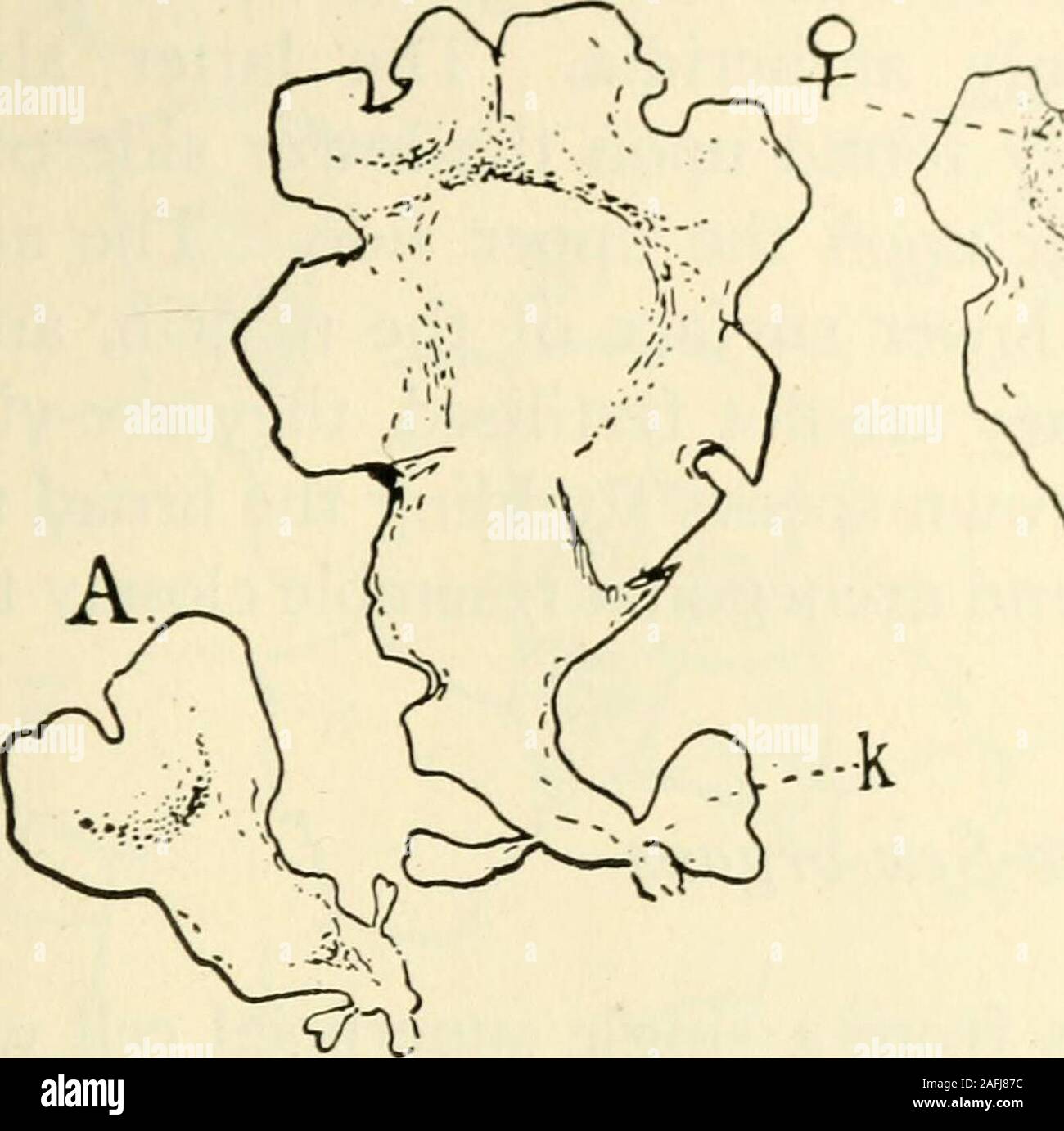
RM2AFJ87C–. The structure and development of mosses and ferns (Archegoniatae). Fig. 150.—Marattia Douglasii. A, Horizon-tal section of prothallium apex, with twoinitials, Xi6o. B, Longitudinal sectionof a similar growing point; d, dorsal; v,ventral segment. VIII MARATTIALES 277 closely that of Angiopteris, than that of Marattia. The rhizoidsare miilticeHular, recalhn^ those of the ^ametophyte ofBotrychinm. The very old prothalHa sometimes hranch dichotomously(Fig. 151, B, C), and the process is identical with tliat in thethallose Hepatic?e. The two growing points are separated bya median lobe in the sam
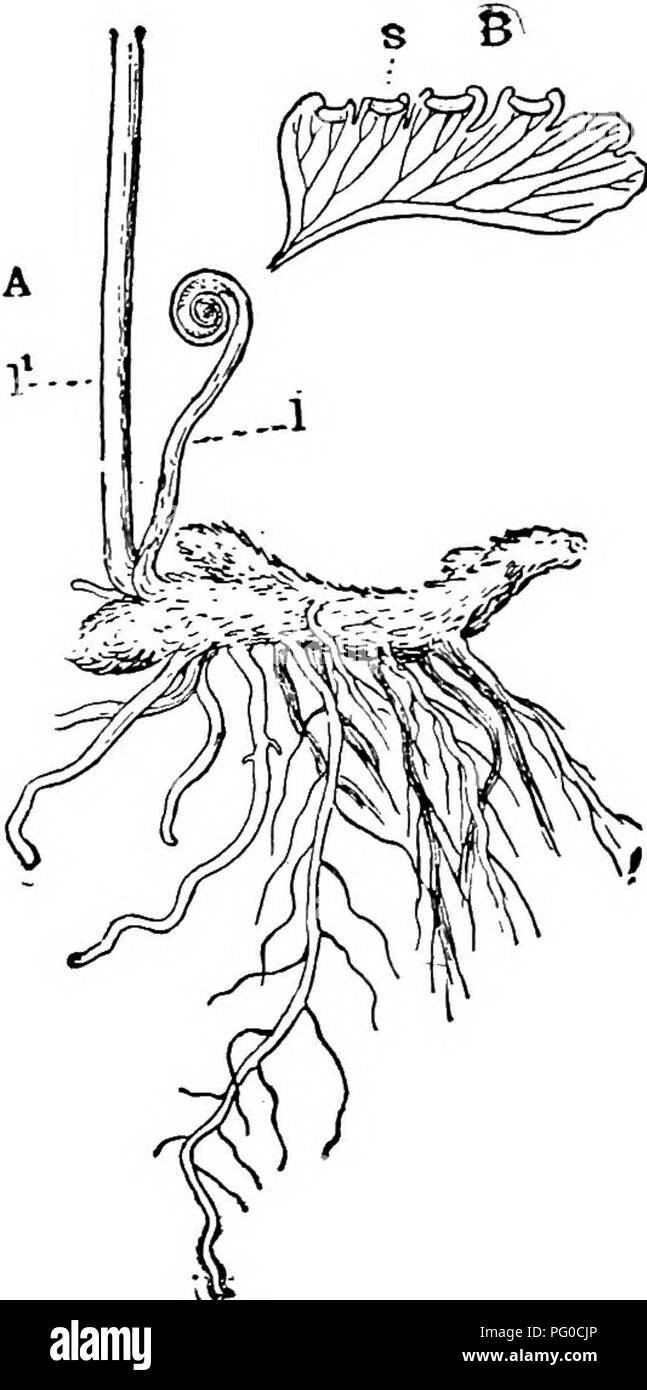
RMPG0CJP–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. i^ FILICINE^ LEPTOSPORANGIATM 327 Marattia. As the stem grows, the central stele, which at first is solid ("protostelic"), becomes a hollow cylinder ("siphonos- tele"), which, according to Jeffrey (3) in most Polypodiaceae shows a concentric structure, i. e., there is a central mass of wood, with both outer and inner phloem, and an external and internal endodermis. Sometimes, however, e. g., Davallia stricta, both internal endodermis and phloem are absent, and this would se
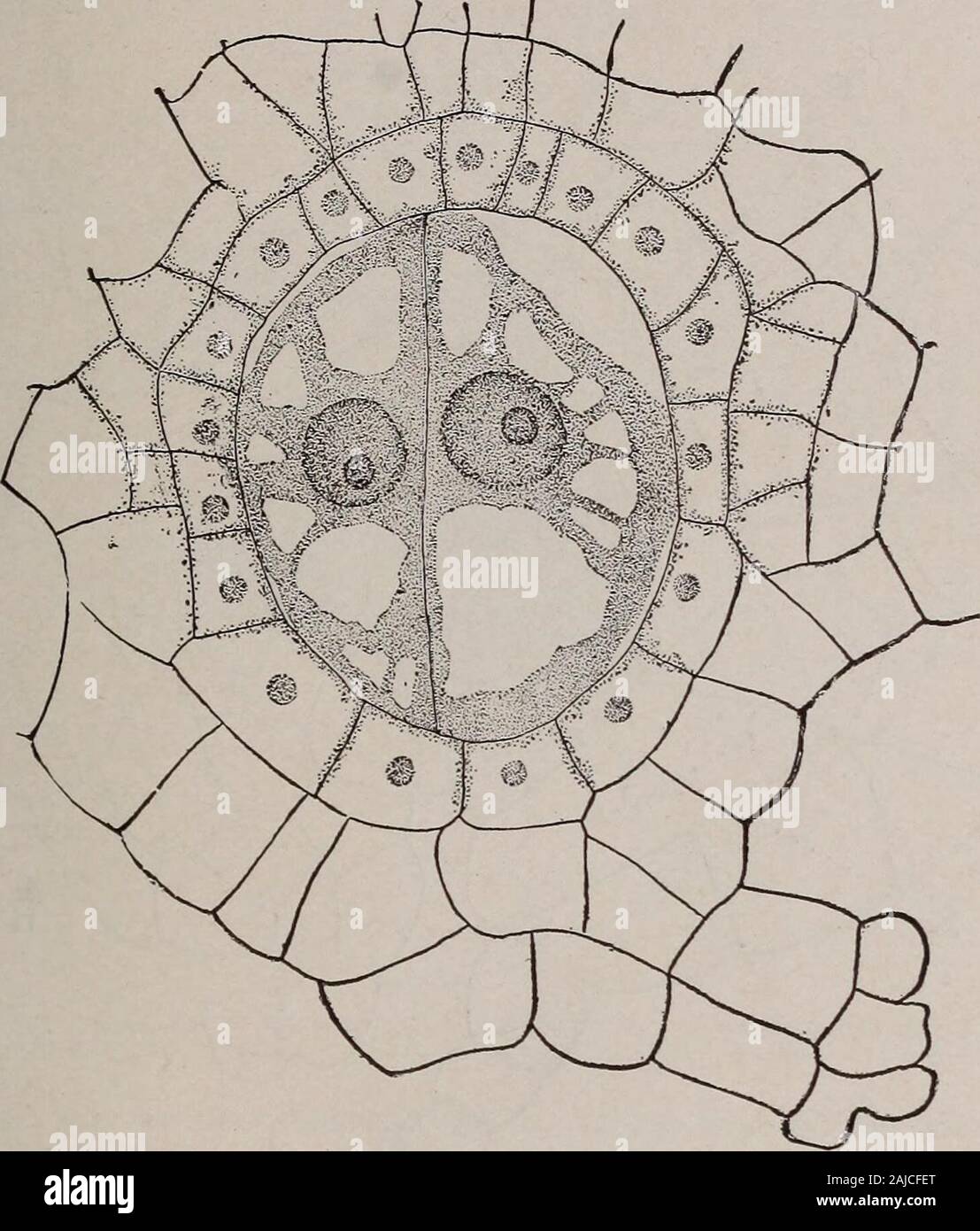
RM2AJCFET–Elementary botany . Fertilization in a fern Marattia). s/>, spermato-zoid fusing with the nu-cleus of the egg. (After Campbell. 1 Sporophyte. 558. Embryo.—The egg first divides into two cells as shown in fig. 228, theninto four. Now from each one of these quandrants of the embryo a definitepart of the plant develops, from one the first leaf, from one the stem, fromone the root, and from the other the organ which is called the foot, and which FERNS. 269 attaches the embryo to the prothallium, and transports nourishment for theembryo until it can become attached to the soil and lead an indepe
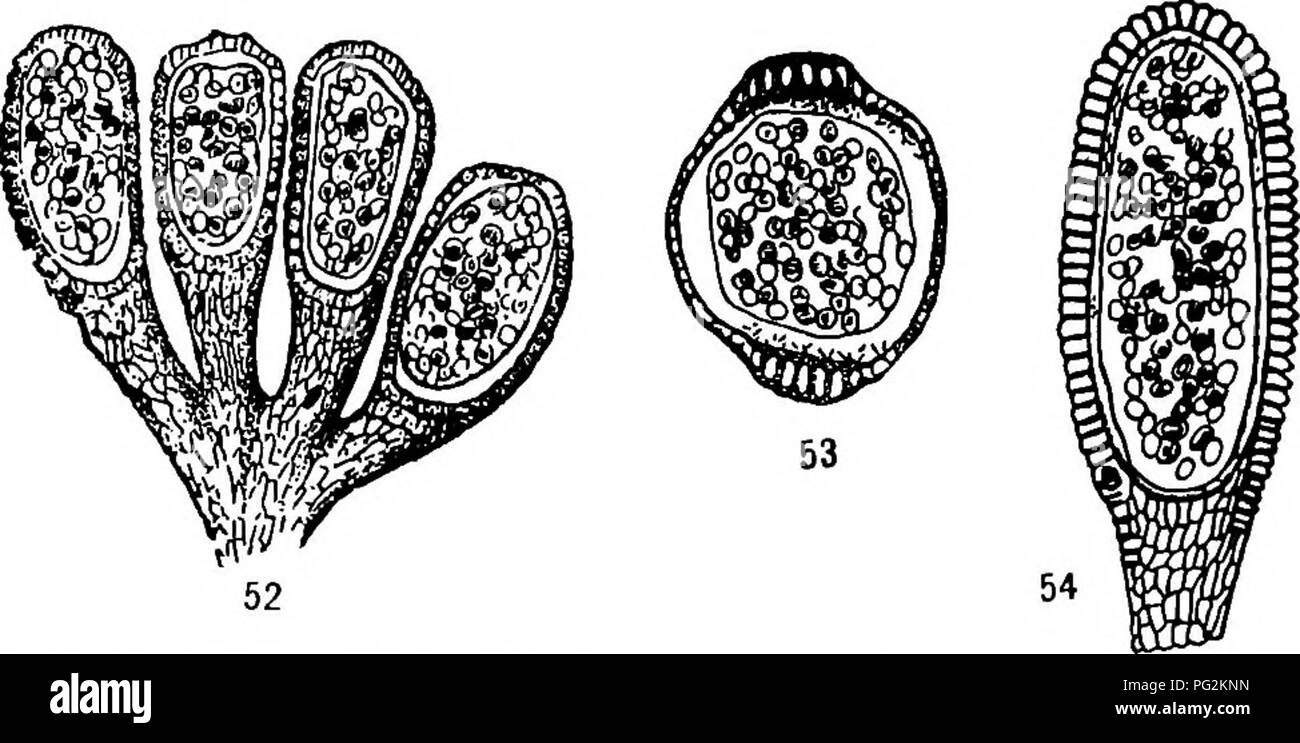
RMPG2KNN–. Morphology of gymnosperms. Gymnosperms; Plant morphology. CYCADOFILICALES 55 have given rise at least to the more modern leptosporangiate and eusporangiate ferns. It is a type older than the paleozoic Marattia- ceae described above, and it is interesting to note that the sporangia were free, rather than in synangia; the conclusion having been drawn (82) that "the fossil evidence, on the whole, supports the view that free sporangia represent the original form of filicinean fructification.. Figs. 52-54.—Zygopteris sp.: fig. 52, four sporangia on a common pedicel; Xio; fig- S3> transver
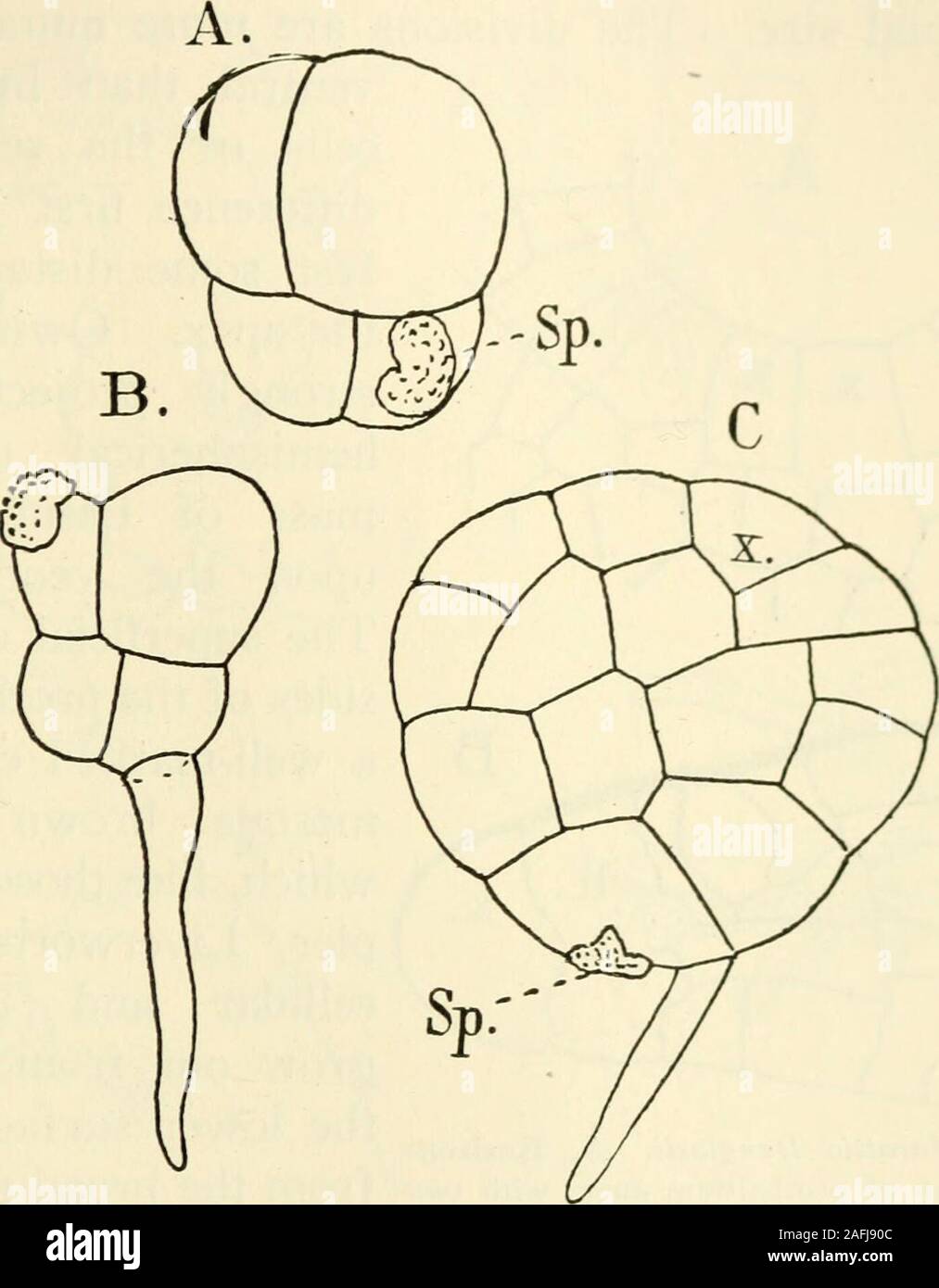
RM2AFJ90C–. The structure and development of mosses and ferns (Archegoniatae). apical cell from whichthe young prothallium for a long time grows (Fig. 149), muchas in Aneura. This type of prothallium, according to Jonkman,is commoner in Marattia than in Angiopteris^ where more com-monly a cell mass is the first result of germination. This latteris usually derived from the form where a rhizoid is developedat first. In this case only the larger of the primary cells givesrise to the prothallium. In the larger cell, divisions take placein three directions and transform it into a nearly globular cell VIII MA
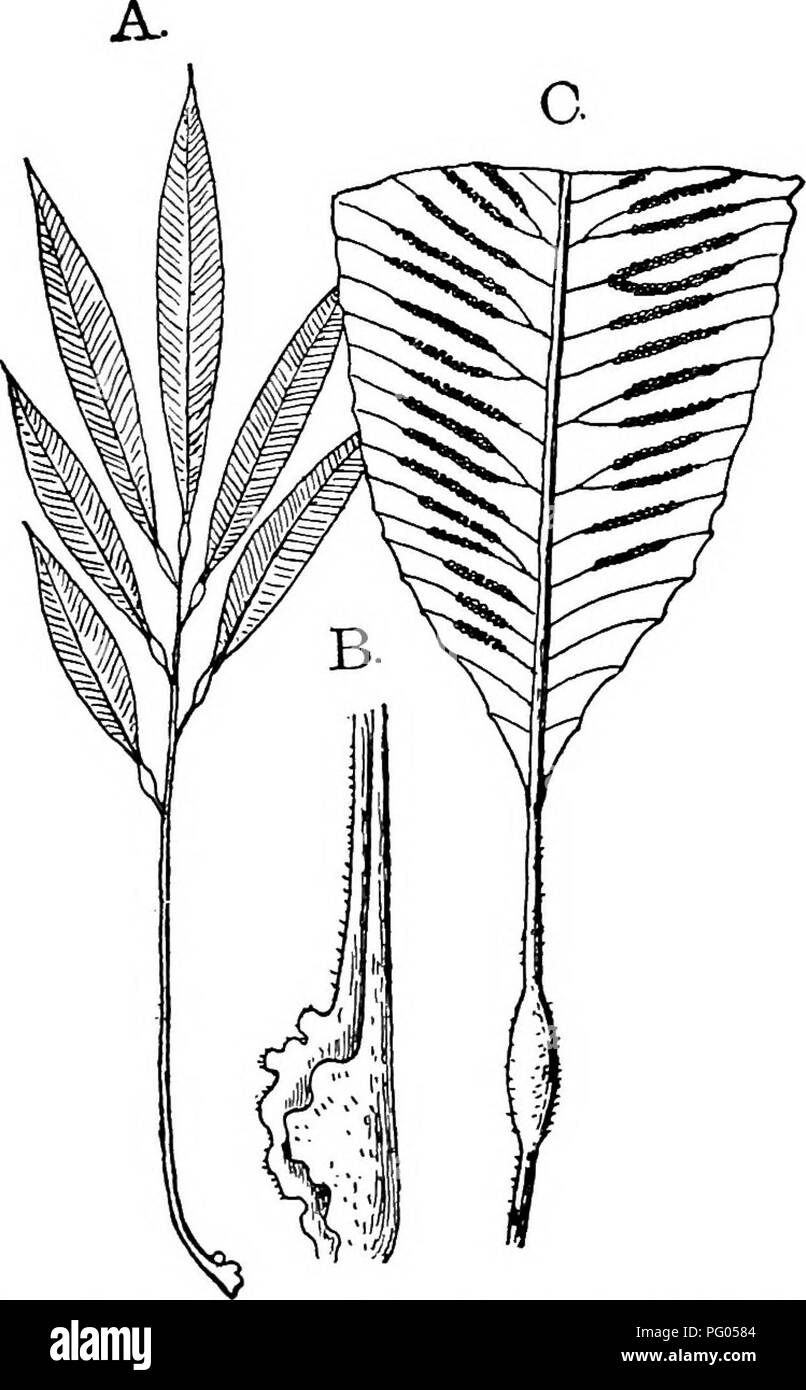
RMPG0584–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. vm MARATTIALES 297 shrinking of the cells on either side of the opening slit. The latter in Dancea is short, and finally appears like a circular pore, but is really not essentially different from that in Kaulfussia and Marattia. In the latter there is a mechanical tissue which causes the two valves of the synangium to gape widely at ma- turity, and the dehiscence of the individual loculi is effected by. Fig. i68.—Archangiopteris Henryi. A, Entire sterile leaf, reduced; B, base of stipe, showin
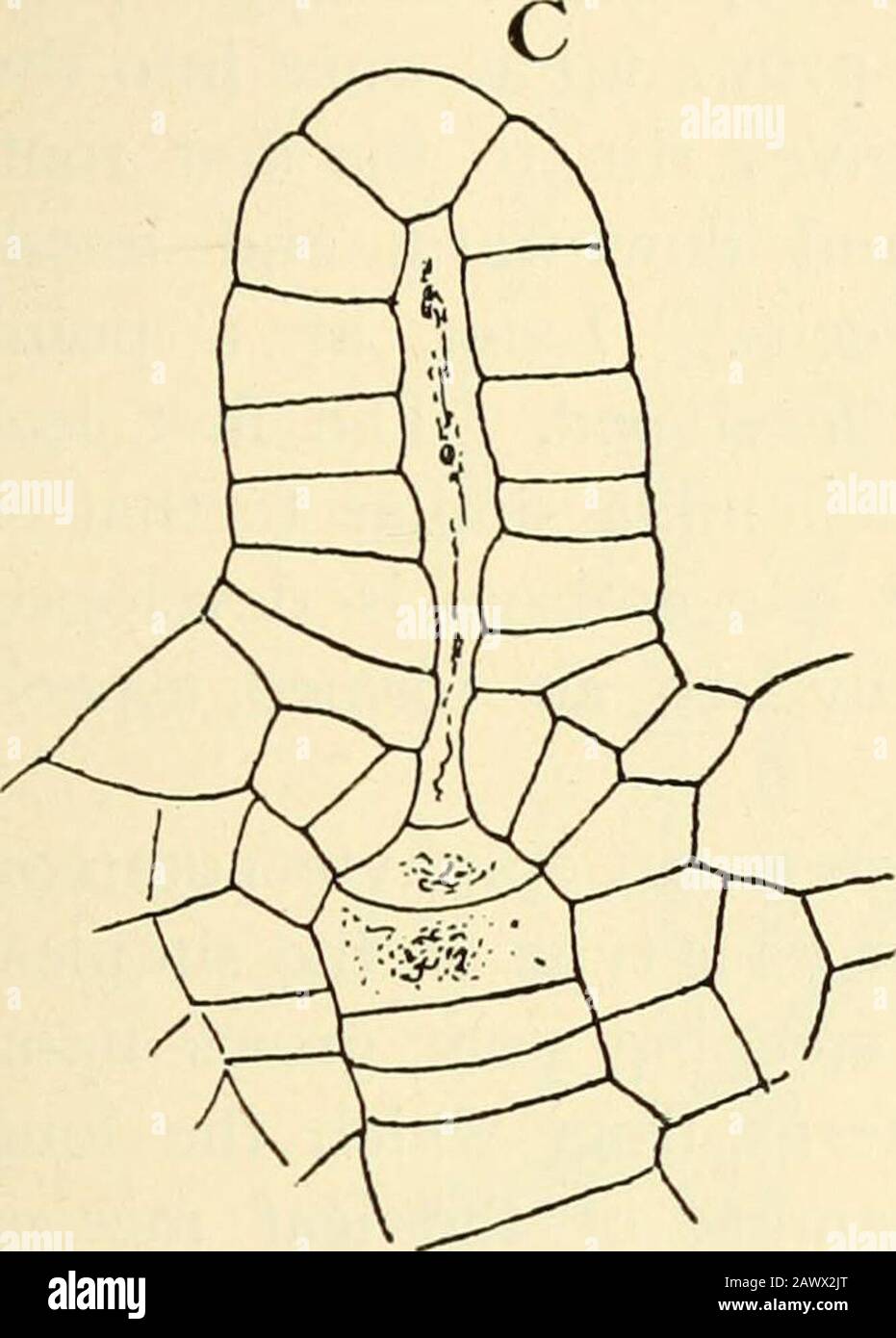
RM2AWX2JT–The structure & development of the mosses and ferns (Archegoniatae) . by vertical and transversewalls, so that the outer wall of the ripe antheridium is two-layered, as in B. lunaria ; but quite as often it remains but onecell thick (Fig. i i i, B), in which respect it resembles Equisetumor Marattia. The spermatozoids were not observed. In only one case was a young embryo found, and this, sofar as could be determined, also resembled in the arrangementof its cells the similar condition in Marattia, but the prepara- VIII THE P TERIDOPH VTA—OF N/0 GL OSS A CE^E 227 tion was not a satisfactory one
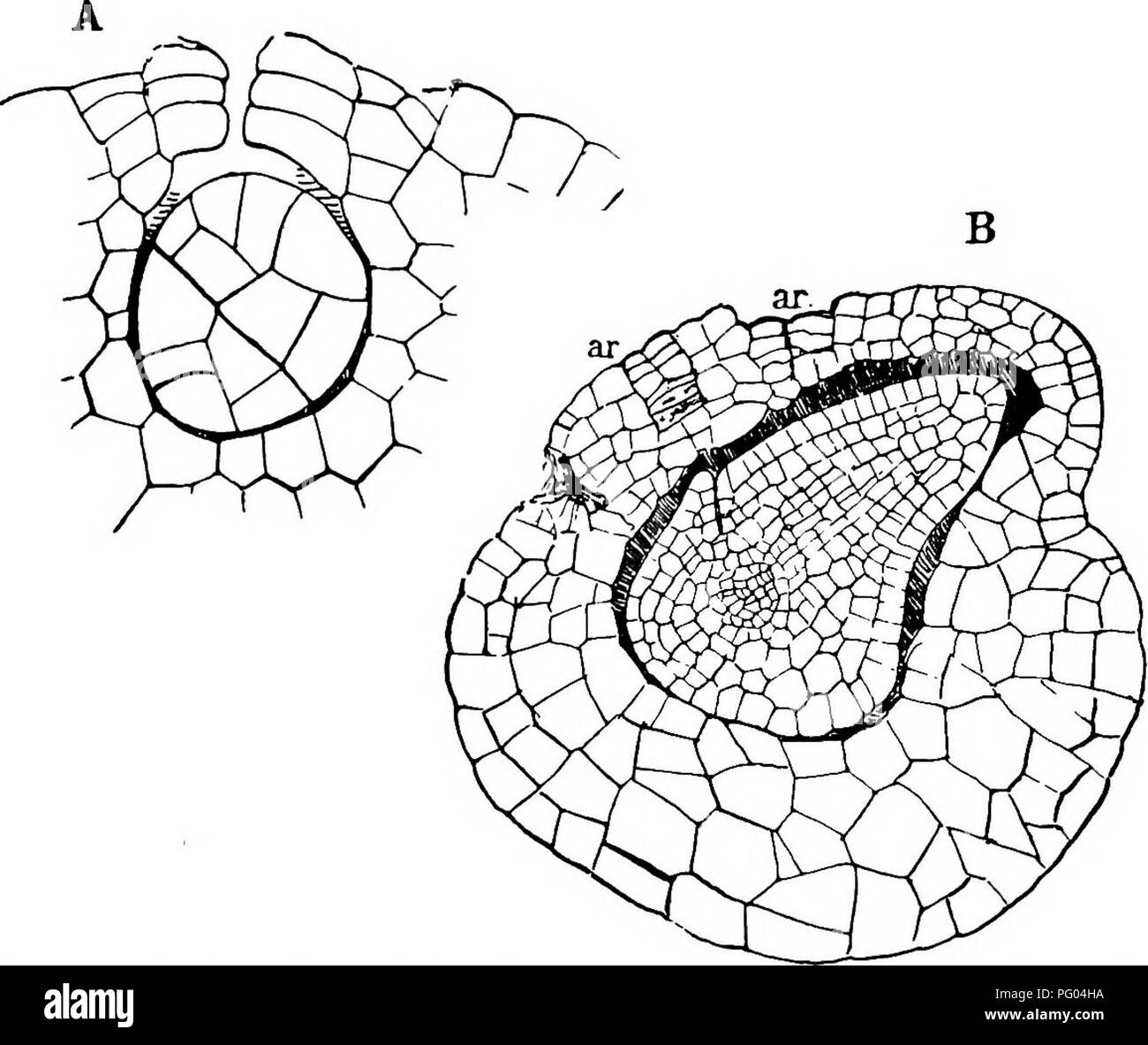
RMPG04HA–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. 546 MOSSES AND FERNS CHAP. but in several young embryos examined, no definite octant walls were present, at least in the upper octants, but whether this is a common occurrence would be difficult to say. The next divisions in the embryo resemble those in Marattia, and as in the latter it may be said that the young members of the embryo grow for a short time from an apical cell, inasmuch as the tetra- hedral octants at first have segments cut off parallel with the basal, quadrant, and octant wal
![The structure & development of the mosses and ferns (Archegoniatae) . Fig. 131.—Marattia Dou^/asii (Ba.]<^er). A, Hori-zontal section of prothallium apex, with twoinitials, Xi6o. B, Longitudinal section of asimilar growing point; d, dorsal; v, ventralsegment. 258 MOSSES AND FERNS CHAP. of the thallus and merges gradually into the wings, which arealso several-layered, nearly or quite to the margin. The very old prothallia sometimes branch dichotomously(Fig. 132, B, C), and the process is identical with that in thethallose Hepaticae. The two growing points are separated bya median lobe in the Stock Photo The structure & development of the mosses and ferns (Archegoniatae) . Fig. 131.—Marattia Dou^/asii (Ba.]<^er). A, Hori-zontal section of prothallium apex, with twoinitials, Xi6o. B, Longitudinal section of asimilar growing point; d, dorsal; v, ventralsegment. 258 MOSSES AND FERNS CHAP. of the thallus and merges gradually into the wings, which arealso several-layered, nearly or quite to the margin. The very old prothallia sometimes branch dichotomously(Fig. 132, B, C), and the process is identical with that in thethallose Hepaticae. The two growing points are separated bya median lobe in the Stock Photo](https://c8.alamy.com/comp/2AWWH6T/the-structure-development-of-the-mosses-and-ferns-archegoniatae-fig-131marattia-douasii-ba-lter-a-hori-zontal-section-of-prothallium-apex-with-twoinitials-xi6o-b-longitudinal-section-of-asimilar-growing-point-d-dorsal-v-ventralsegment-258-mosses-and-ferns-chap-of-the-thallus-and-merges-gradually-into-the-wings-which-arealso-several-layered-nearly-or-quite-to-the-margin-the-very-old-prothallia-sometimes-branch-dichotomouslyfig-132-b-c-and-the-process-is-identical-with-that-in-thethallose-hepaticae-the-two-growing-points-are-separated-bya-median-lobe-in-the-2AWWH6T.jpg)
RM2AWWH6T–The structure & development of the mosses and ferns (Archegoniatae) . Fig. 131.—Marattia Dou^/asii (Ba.]<^er). A, Hori-zontal section of prothallium apex, with twoinitials, Xi6o. B, Longitudinal section of asimilar growing point; d, dorsal; v, ventralsegment. 258 MOSSES AND FERNS CHAP. of the thallus and merges gradually into the wings, which arealso several-layered, nearly or quite to the margin. The very old prothallia sometimes branch dichotomously(Fig. 132, B, C), and the process is identical with that in thethallose Hepaticae. The two growing points are separated bya median lobe in the

RMPG01DR–. A natural history of new and rare ferns : containing species and varieties, none of which are included in any of the eight volumes of "Ferns, British and exotic", amongst which are the new hymenophyllums and Trichomanes . Ferns. Portion of fertile Frond, under side. MARATTIA KAULFUSSII. KUNZE. PLATE XVII. Marattia Icevis, Katti,fit38. Martens and Galleotti. Link. (iVbi! of Smith.) " Weinmannicrfolia, Lbibmann. Eupodium Kaulfiissii, J. Smith. Hookbe. Marattia—Named in honour of J. F. Maratti, a Tuscan botanist. Kaulfussii—^ISTamed after the celebrated oryptogamist, Kaulfuss. In
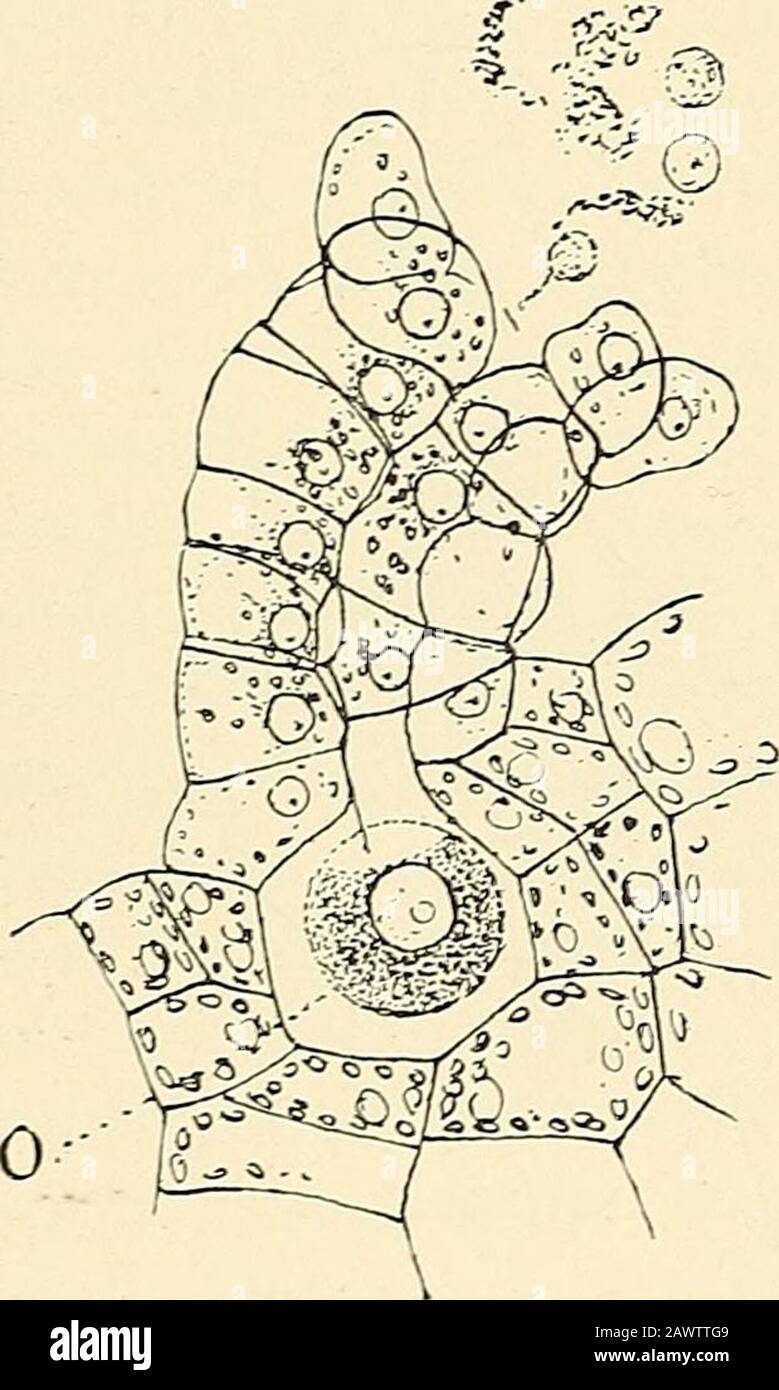
RM2AWTTG9–The structure & development of the mosses and ferns (Archegoniatae) . ntralcells. The cover cell divides intothe four primary neck cells, and thecentral cell arching up between thesehas the pointed apex cut off by acurved wall from the central cell.The primary neck canal cell, soformed, is noticeably smaller thanthat of Marattia. The neck cells,which in the eusporangiate forms allgrow alike, here show a difference,and the two anterior rows developfaster than the posterior ones, so that these rows are longer andthe neck is strongly bent backward. In Onoclea there areusually about seven cells in
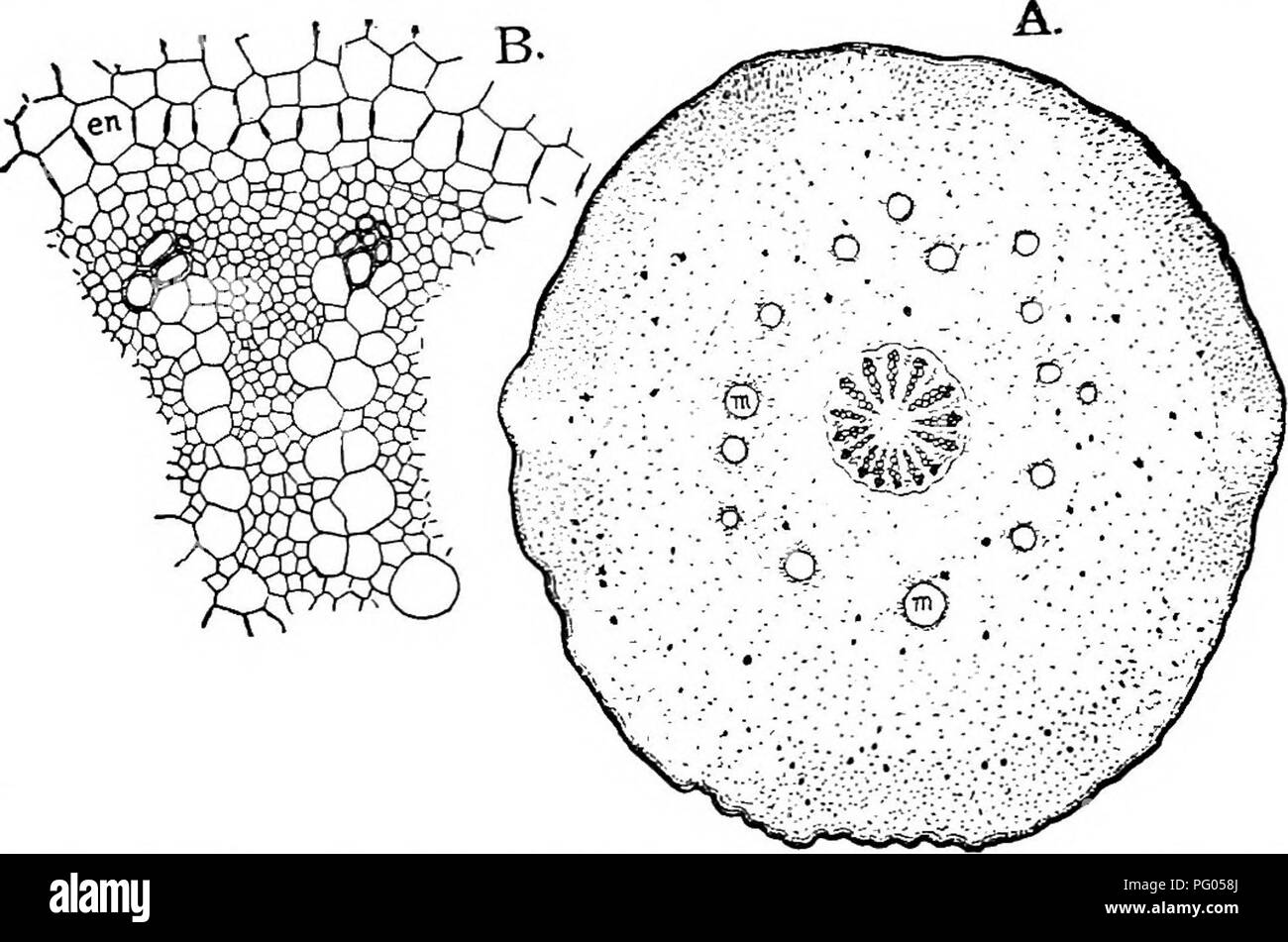
RMPG058J–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. VIII MARATTIALES 291 ever, bath of Marattia and Angiopteris, there is but a single axial bundle, as in the petiole of the cotyledon. Fig. 167, B shows a cross-section of a pinnule from a large leaf of A. evecta, which has much the same structure as that of Marattia. The central vascular bundle is horse-shoe shaped in section, and shows a central mass of large tracheids with retic- ulate or scalariform markings, surrounded by the phloem made up of very large sieve-tubes much like those of Botry
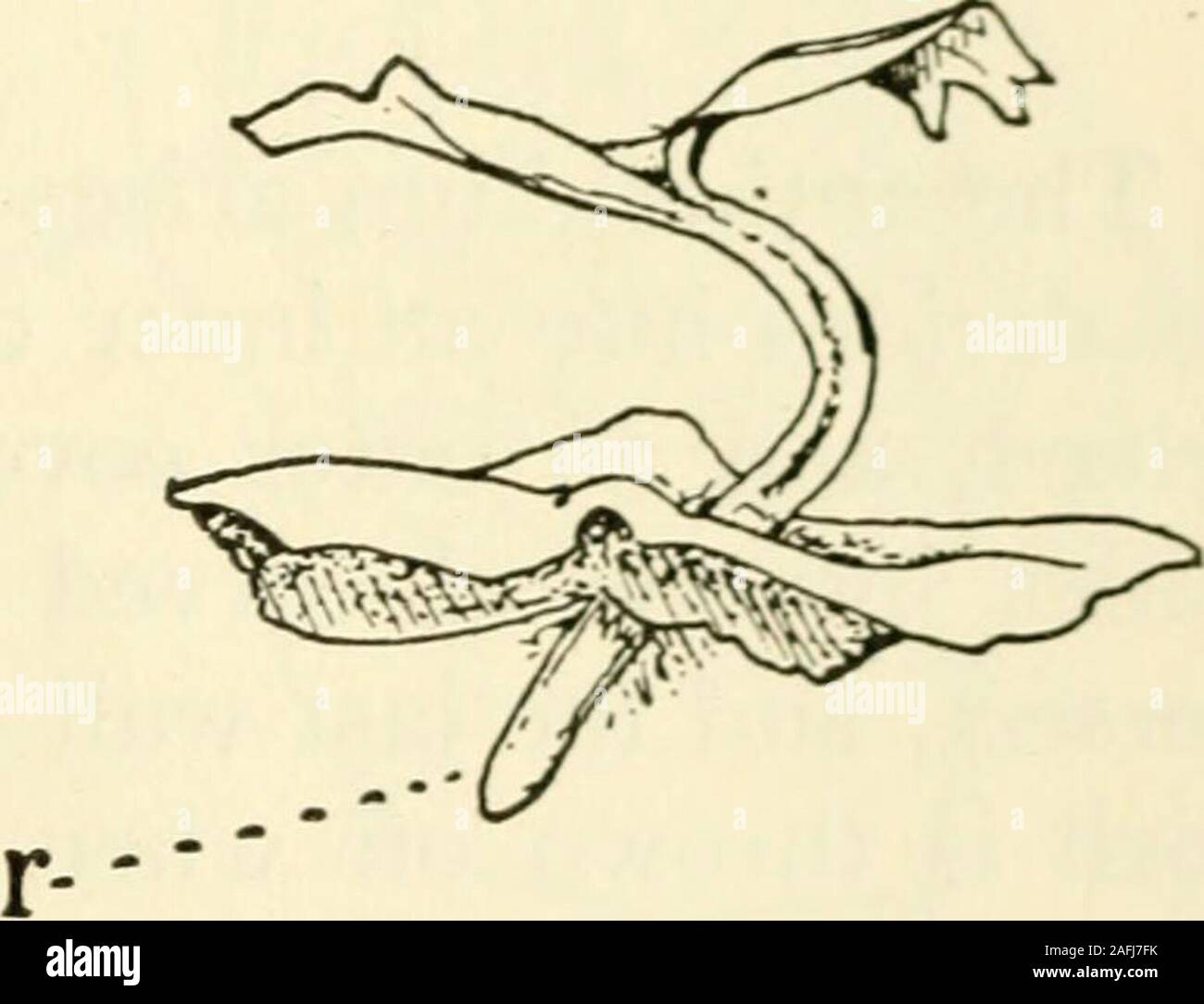
RM2AFJ7FK–. The structure and development of mosses and ferns (Archegoniatae). Fig. 151.—Marattia Douglasiu A, Prothallium about one year old, X2; B, the sameprothallium about a year later, showing a dichotomy of the growing point; C, thesame seen from below, showing two archegonial cushions (^); D, prothallium withyoung sporophyte, X4; E, a somewhat older one, seen from the side; r, the pri-mary root. organs upon It forks with it, exactly as we find, for example,the antheridial receptacle forking in Fimbriaria Californica(Fig. I, A). Besides this form of branching, which is notcommon, adventitious buds
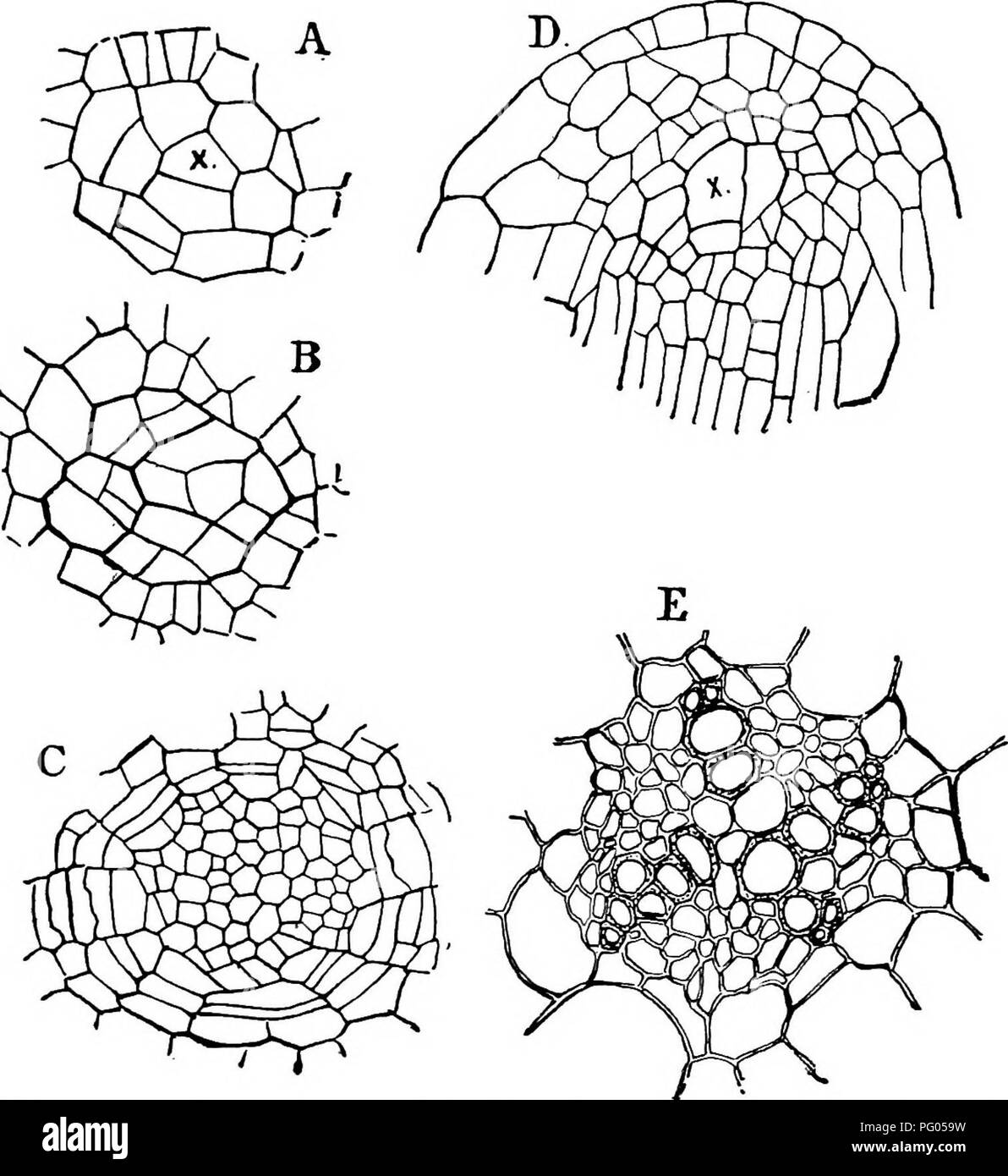
RMPG059W–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. Vlll MARATTIALES 283 At first the growth is nearly vertical, but it soon becomes stronger upon the outer side, and the leaf rudiment bends inwards. At this stage the different tissues begin to be dis- tinguishable. Somewhat later the tip of the cotyledon becomes flattened, and still later there is a dichotomy of this flattened part which thus forms a fan-shaped lamina (Fig. 157). The. Fig. 156.—Marattia Douglasii. A, B, C, Three transverse sections of a root from the young sporophyte; A shows
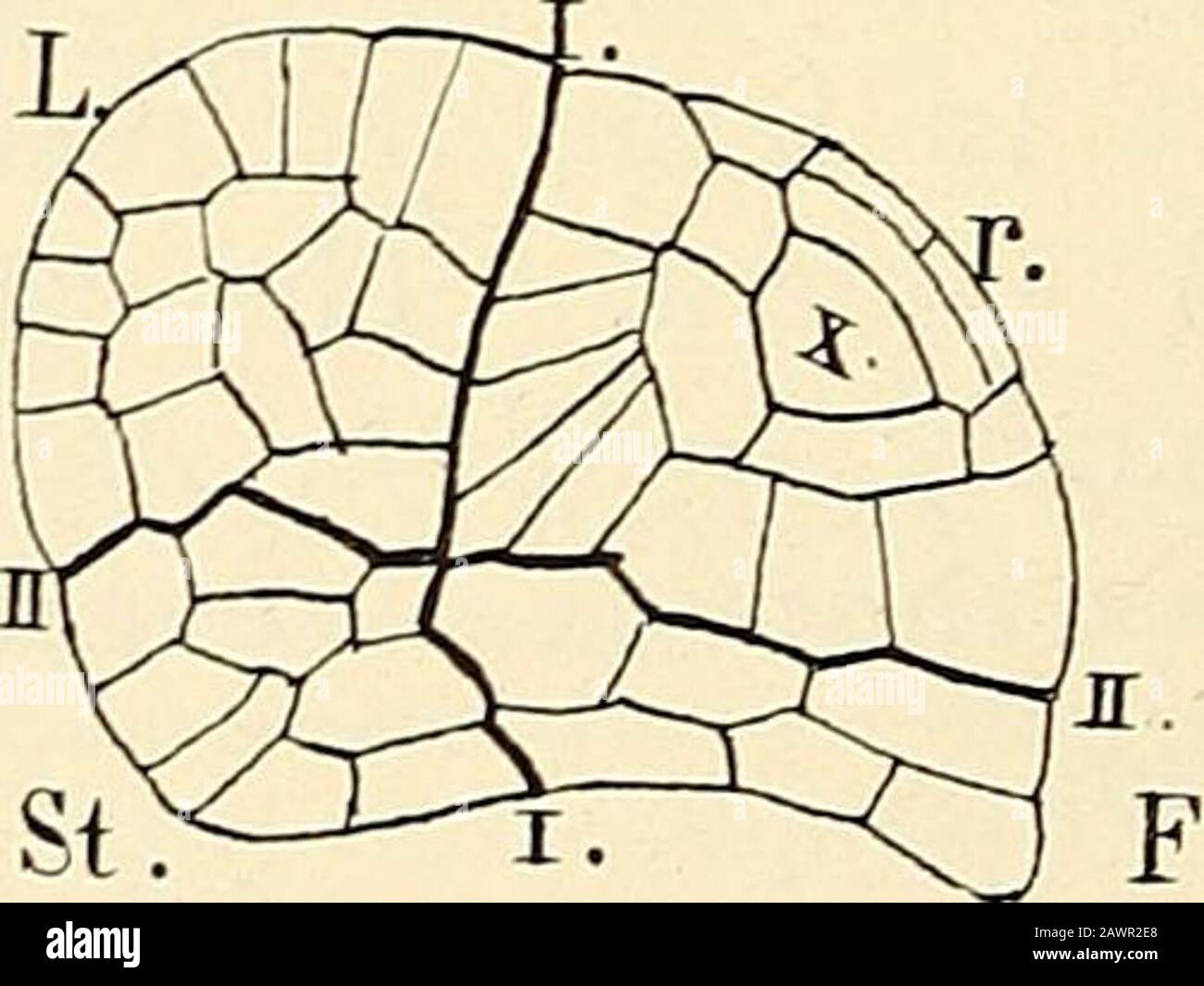
RM2AWR2E8–The structure & development of the mosses and ferns (Archegoniatae) . Jig. 212.—Marsilia vestita (Hook and Grev.). Development of the embryo. A, Longitudinalsection of archegonium with two-celled embrjo; B, similar section of a later stage ; C, twotransverse sections of a young embryo ; D, two longitudinal sections of an older one; I, I, thebasal wall; L, cotyledon ; st, stem; r, root ; F, foot. A-C, X 525 ; D, X260. the changes are the same as those described in Marattia andOsmunda. Coincident with the first divisions in the embryo, eachof the lateral cells of the prothallium (venter) divides
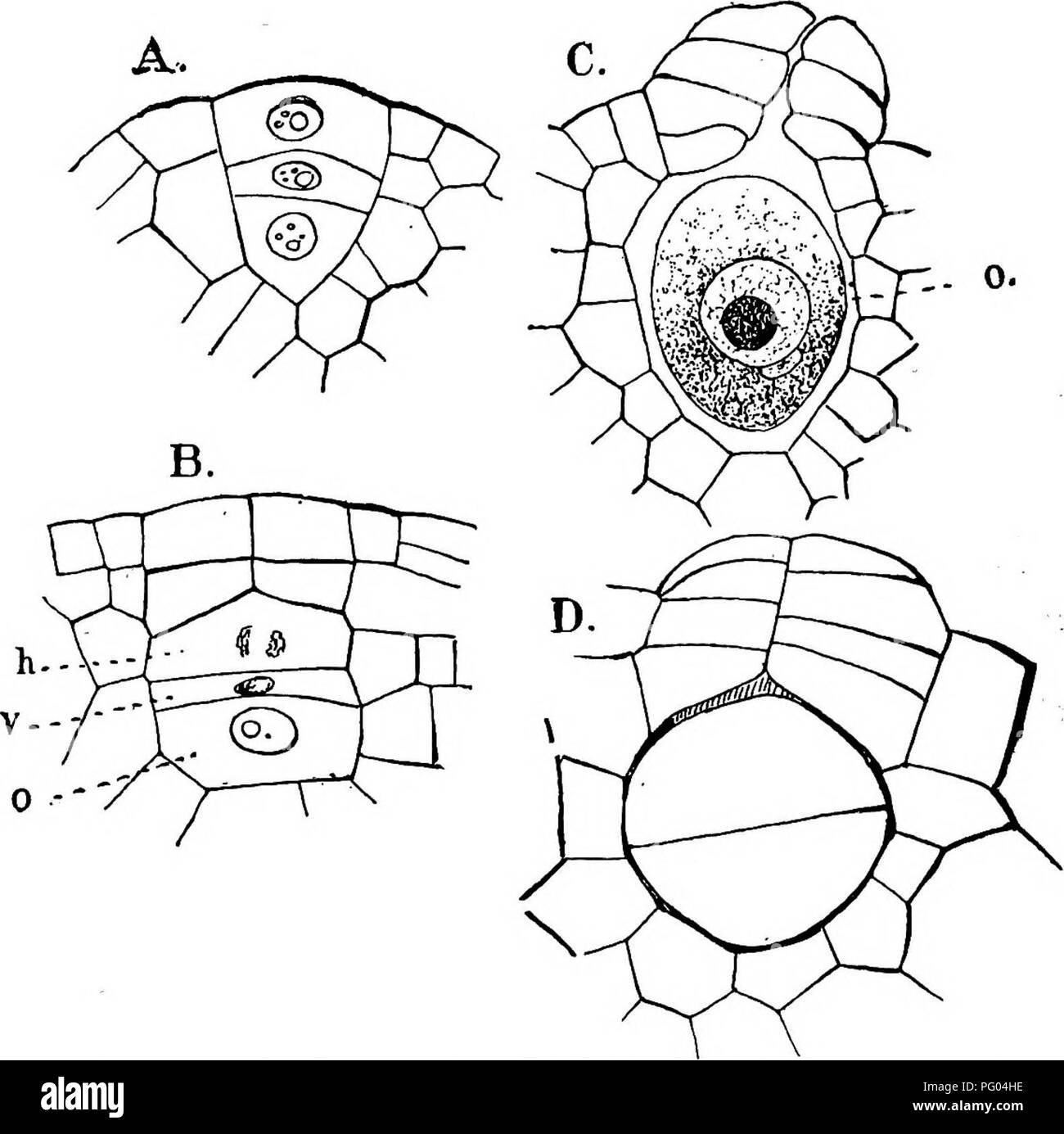
RMPG04HE–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. S44 MOSSES AND FERNS CHAP. than in Marattia, the neck canal cell is shorter and extends but little between the neck cells (Fig. 313, B). The e.gg is very large, round or oval in form, and the nucleus contains a large nucleolus that stains very intensely, but otherwise shows little chromatin. The receptive spot is of unusual size, and occupies about one-third of the &gg. It is. Fig. 3ii.—Isoeies eehinospora var. Braunii. Development of the archegonium, X500; 0, the e^g', V, ventral canal ce
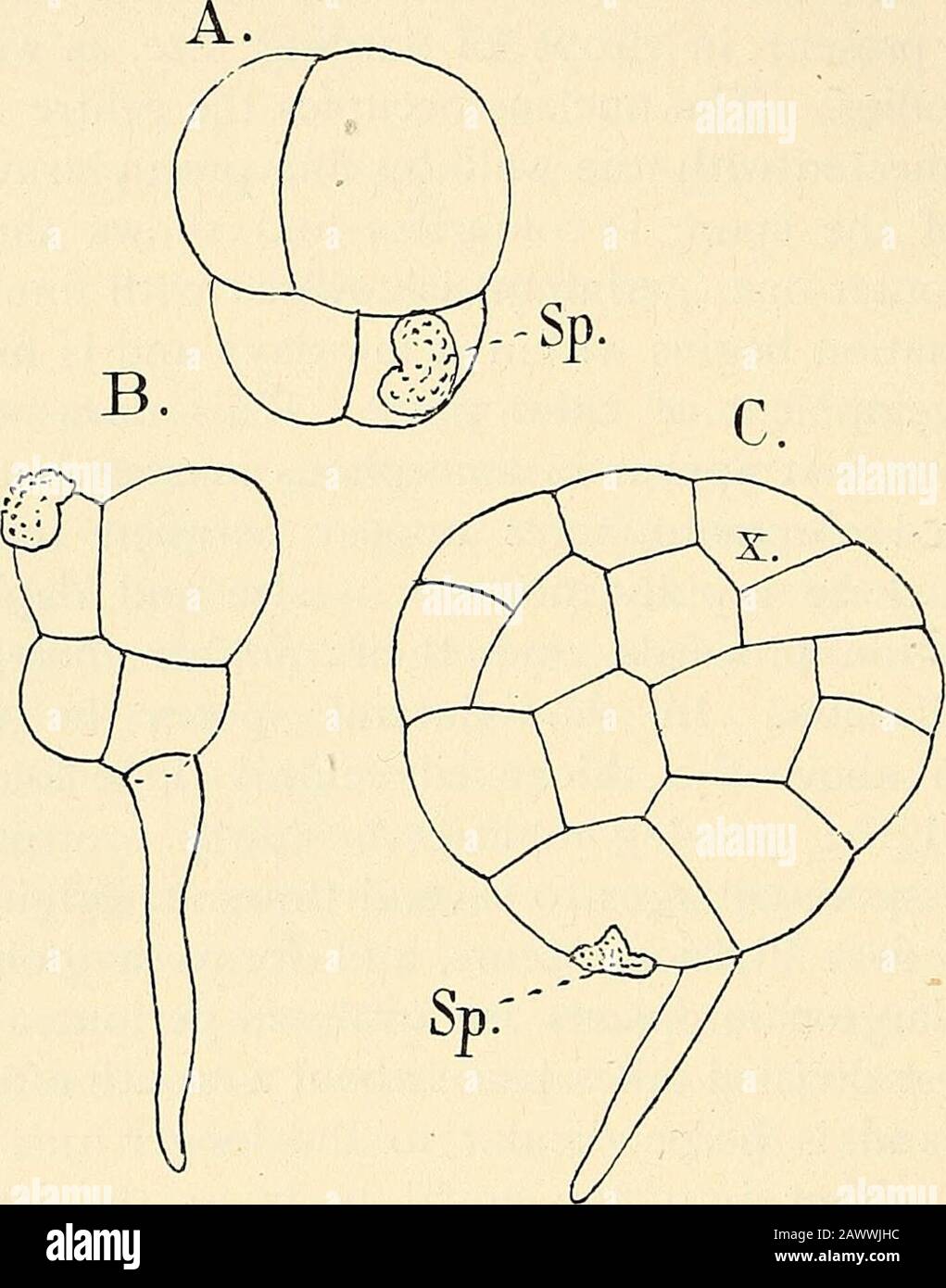
RM2AWWJHC–The structure & development of the mosses and ferns (Archegoniatae) . the two-sided apical cell from whichthe young prothallium for a long time grows (Fig. 130), muchas in Aneiira. This type of prothallium, according to Jonkman,^is commoner in Marattia than in Angiopteris, where morecommonly a cell mass is the first result of germination. Thislatter is usually derived from the form where a root-hair isdeveloped at first. In this case only the larger of the primary 1 Luerssen (5). ^ Jonkman (i). ^ Jonkman (2). ?* Jonkman, Bot. Zcit. 1S7S, p. 136. ^ Jonkman, I.e. p. 146. 256 MOSSES AND FERNS CHA
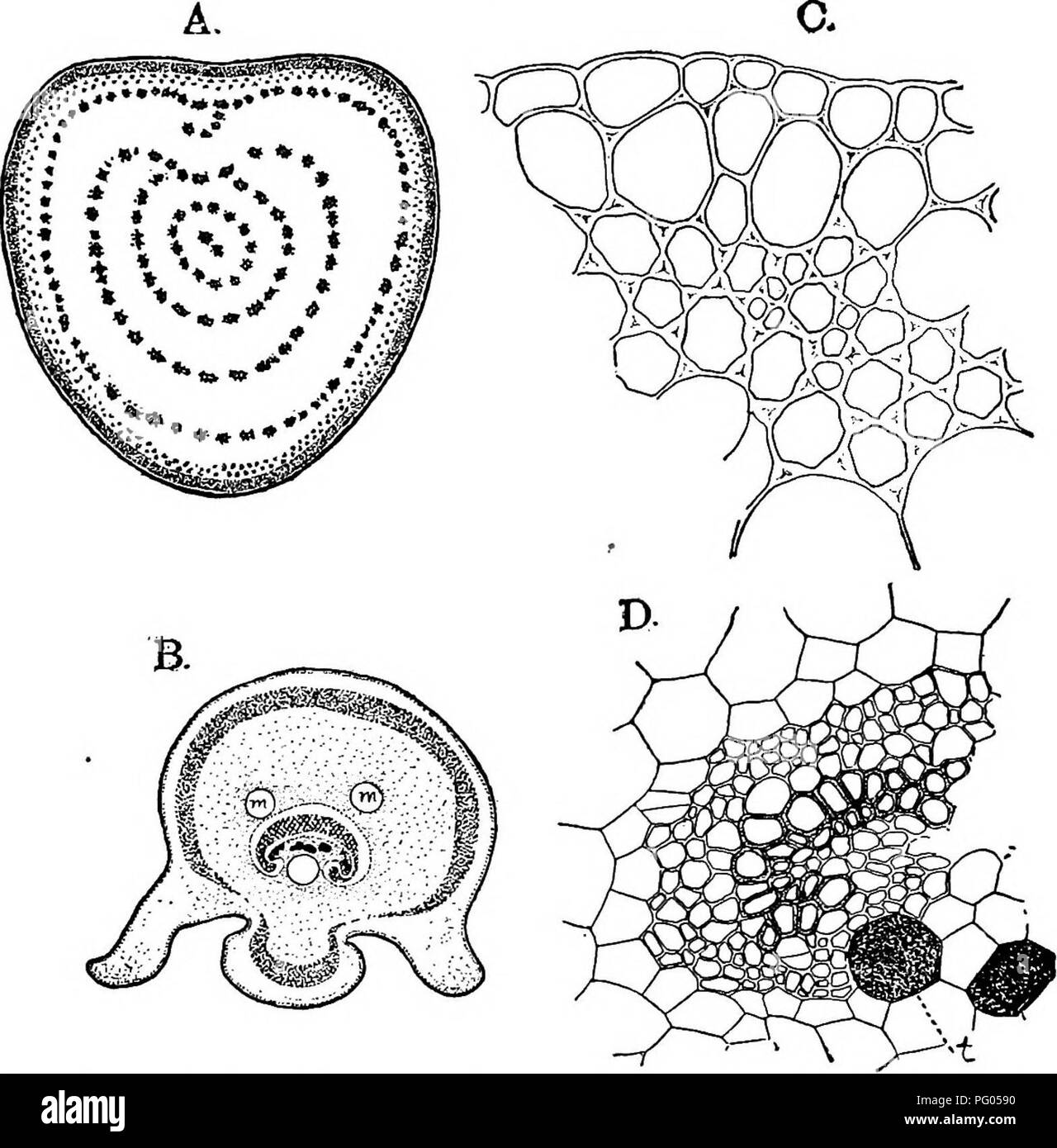
RMPG0590–. The structure and development of mosses and ferns (Archegoniatae). Plant morphology; Mosses; Ferns. vin MARATTIALES 289 The Adult Sporophyte According to Holle (1. c. p. 218) the four-sided apical cell found in the stem of the young sporophyte of Marattia is re- tained permanently, but in Angiopteris this is not the case, as in the older sporophyte a single apical cell is not certainly to be made out. Bower ((11) p. 324) comes to the same conclusion. Fig. 161.—A, Section of the stipe of Angiopteris evecta, natural size; B, section of the rachis of the ultimate division of the leaf of Maratti
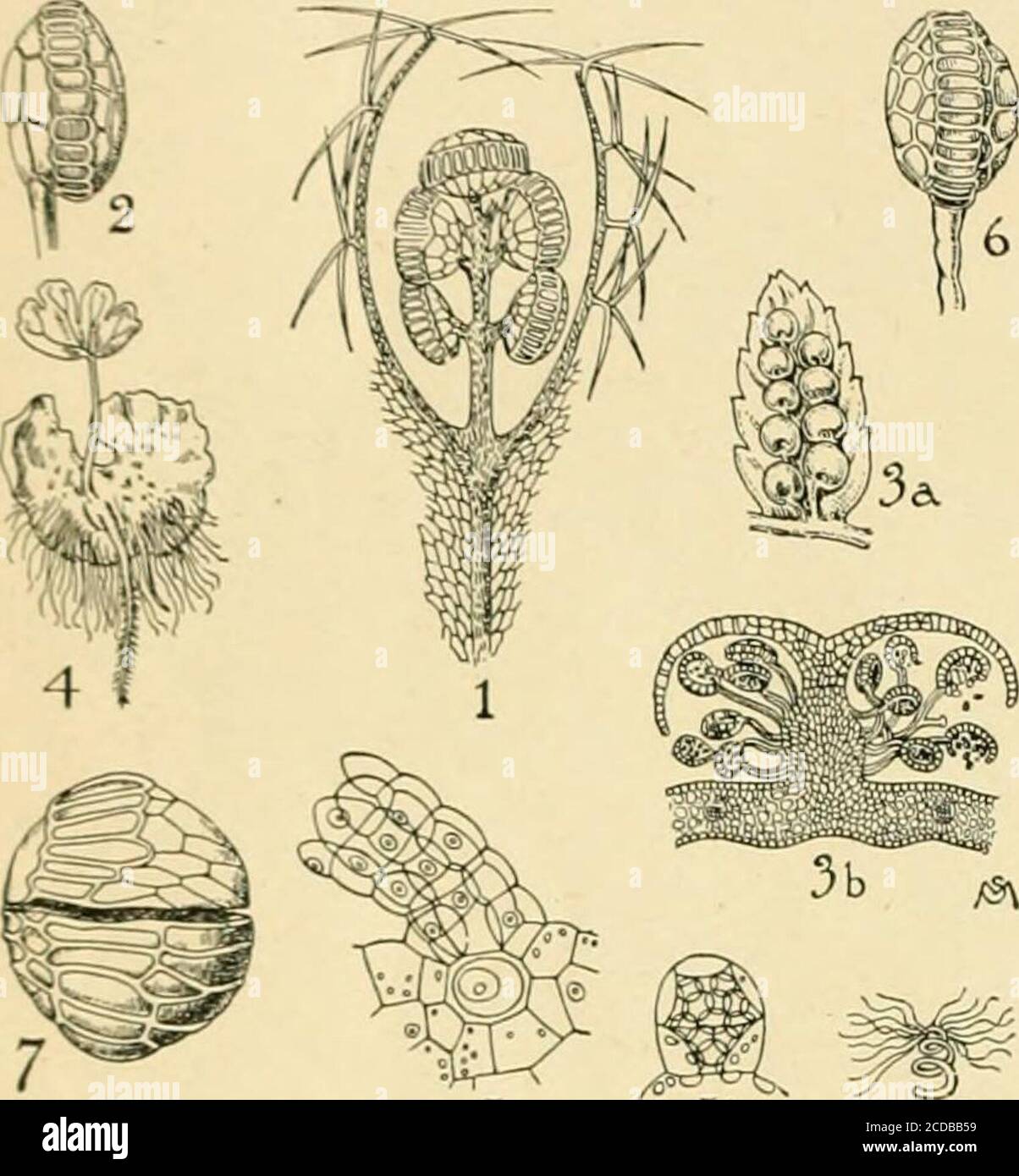
RM2CDBB59–. The standard cyclopedia of horticulture; a discussion, for the amateur, and the professional and commercial grower, of the kinds, characteristics and methods of cultivation of the species of plants grown in the regions of the United States and Canada for ornament, for fancy, for fruit and for vegetables; with keys to the natural families and genera, descriptions of the horticultural capabilities of the states and provinces and dependent islands, and sketches of eminent horticulturists . od. The fleshy stipulesof Marattia fraxinea are eaten; the spicy leaves of somespecies are used to season
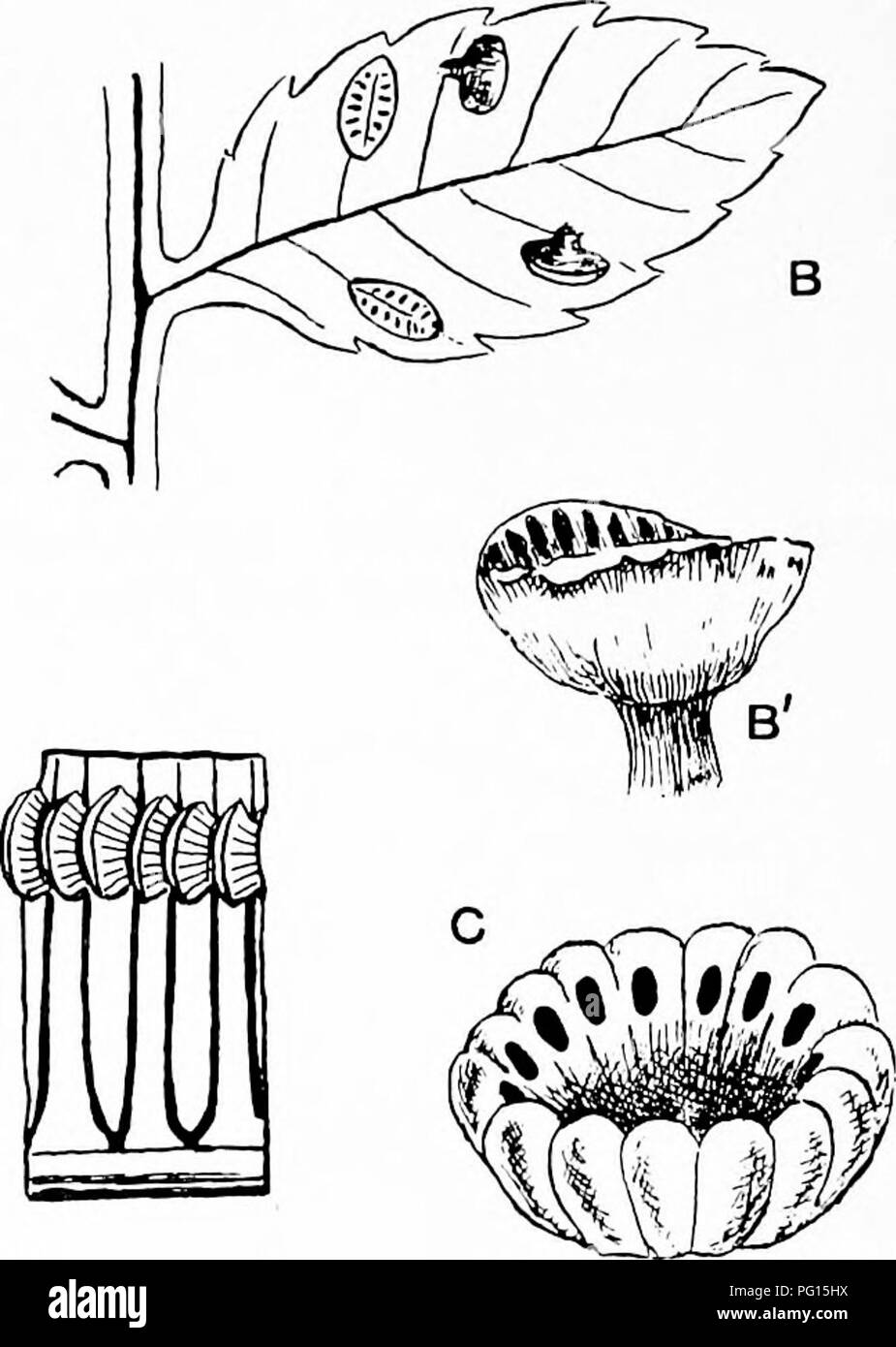
RMPG15HX–. Fossil plants : for students of botany and geology . Paleobotany. Fig. 245. A. Marattia fraxinea. A'. A single synangium showing the two valves and pores of the sporangial compartments. B, B'. it/. Kaulfussii. C, Kaulfussia (synangium showing pores of sporangial compart- ments). D, B. Marattiopsis Milnsteri. (C, after Hooker; D, E, after Schimper.) composed of two valves, which on ripening come apart and expose two rows of pores formed by the apical dehiscence of the sporangial com- partments (fig. 245, A', B). In Marattia Kaulfussii the sori are attached to the lamina by a short stalk (fig.
RMRMWH9K–. Annales des Sciences Naturelles Botaniques. 220 «ËORCiEii POIRAULiT. maria Patersoni^ Platycerium alcicorne, Polypodium decur- rens, Marattïa Brongniartii, Angiopteris Durvilleana, etc. (fig. 32).. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Paris

RMPG15K5–. Fossil plants : for students of botany and geology . Paleobotany. Fio. 241. A. Angiopteris evecta. (Cousiderablj' reduced.) B. Marattia fraxinea. Stipule. M.S. elliptical sori near the edge of the pinnules, consist of free sporangia (fig. 242, A—D) provided with a peculiar type of "annulus "2, in the form of a narrow band of thicker-walled cells, which extends as a broad strip on either side of the apex. An examination of sections through the sporangia of Angiopteris in different planes'' illustrates the difficulty of determining the precise nature of the annulus in a petrified spo
Search Results for Marattia Stock Photos and Images (208)
Page 1 of 3































